1,2-二氧杂环戊烷
外观
(重定向自1,2-二氧雜環戊烷)
| 1,2-二氧杂环戊烷 | |
|---|---|
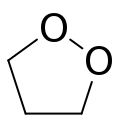
| |
| IUPAC名 1,2-Dioxolane 1,2-二氧戊环 | |
| 系统名 1,2-Dioxacyclopentane 1,2-二氧杂环戊烷 | |
| 别名 | 1,2-二氧五环 1,2-二𫫇环戊烷 |
| 识别 | |
| CAS号 | 4362-13-4 |
| PubChem | 138198 |
| ChemSpider | 121835 |
| SMILES |
|
| 性质 | |
| 化学式 | C3H6O2 |
| 摩尔质量 | 74.08 g·mol−1 |
| 若非注明,所有数据均出自标准状态(25 ℃,100 kPa)下。 | |
1,2-二氧杂环戊烷,别名1,2-二氧戊环、1,2-二氧五环等,是一种化合物,分子式为C3H6O2,由三个碳原子和相邻位置的两个氧原子组成的环组成。其结构式可写为[–(CH2)3–O–O–]。[1][2]
该化合物是一种有机过氧化物,特别是内过氧化物,是更常见的1,3-二氧戊环的结构异构体。
合成
[编辑]1,2-二氧戊环核心结构的合成方法包括用单线态氧[3]或分子氧与合适的催化剂氧化环丙烷衍生物、[4][5]使用自动氧化、过氧化氢进行亲核置换,并用硝酸汞处理、扩展π-系统的光解、[6]双甲硅烷基过氧化物与烯烃的反应,[7]或与2-过羟基4-烯烃与二乙胺[8]或乙酸汞的反应。[9][10]
出现
[编辑]一些衍生物存在于自然界,例如在Calophyllum dispar和马米杏的种子。[6]Plakinic acid A和类似化合物是从Plakortis的海绵中分离出来的。[11][12]甘松新酮是一种含有1,2-二氧戊环的倍半萜衍生物,从名为Adenosma caeruleum的植物中分离出来。[13]
用途
[编辑]合成和天然的二氧戊环衍生物已被用作或被认为是抗疟药。[6][7]Plakinic acid A和相关化合物显示出抗真菌作用。[11]
参见
[编辑]参考资料
[编辑]- ^ Lay TH, Bozzelli JW. Enthalpies of formation of cyclic alkyl peroxides: Dioxirane, 1,2-dioxetane, 1,2-dioxolane, and 1,2-dioxane. Chemical Physics Letters. 199, 268 (1–2): 175–179. Bibcode:1997CPL...268..175L. doi:10.1016/S0009-2614(97)00168-1.
- ^ Demaison J. 2.5.2.5 Elements with two or three internal rotors. Diamagnetic Molecules. Landolt-Börnstein - Group II Molecules and Radicals 14a. 1982: 415–425. ISBN 3-540-11365-7. doi:10.1007/10201404_40.
- ^ Akasaka T, Fukuoka K, Ando W. Formation of 1,2-dioxolane in the singlet oxygenation of cyclopropane. Tetrahedron Letters. 1991, 32 (52): 7695–7698. doi:10.1016/0040-4039(91)80568-Q.
- ^ Feldman KS, Simpson RE. Stereochemical studies on the preparation and subsequent reductive cleavage of 1,2-dioxolanes. Application to the synthesis of (±)-yashabushitriol. Tetrahedron Letters. 1989, 30 (50): 6985–6988. doi:10.1016/S0040-4039(01)93404-5.
- ^ Mata S, González J, Vicente R, López LA. Zinc-Catalyzed Multicomponent Reactions: Easy Access to Furyl-Substituted Cyclopropane and 1,2-Dioxolane Derivatives (PDF). European Journal of Organic Chemistry. 2016, 2016 (15): 2681–2687 [2023-04-26]. doi:10.1002/ejoc.201600393. (原始内容存档 (PDF)于2023-04-26).
- ^ 6.0 6.1 6.2 Helesbeux JJ, Peyronnet D, Labaïed M, Grellier P, Frappier F, Seraphin D, et al. Synthesis and antimalarial activity of some new 1,2-dioxolane derivatives. Journal of Enzyme Inhibition and Medicinal Chemistry. December 2002, 17 (6): 431–7. PMID 12683680. doi:10.1080/1475636021000005677
 .
.
- ^ 7.0 7.1 Martyn DC, Ramirez AP, Beattie MJ, Cortese JF, Patel V, Rush MA, et al. Synthesis of spiro-1,2-dioxolanes and their activity against Plasmodium falciparum. Bioorganic & Medicinal Chemistry Letters. December 2008, 18 (24): 6521–4. PMID 18993067. doi:10.1016/j.bmcl.2008.10.083.
- ^ Liu HH, Jin HX, Wu YK. Synthesis and cleavage studies of a 1,2-dioxolane-type peroxide. Chinese Journal of Chemistry. 2010, 22 (9): 1029–1033. doi:10.1002/cjoc.20040220930.
- ^ Booldworth AJ, Tallant NA. 1,2,4-Trioxane versus 1,2-dioxolane formation in the mercury(II) acetate-mediated cyclisation of hemiperoxyacetals derived from allylic hydroperoxides. Journal of the Chemical Society, Chemical Communications. 1992, (5): 428. doi:10.1039/C39920000428.
- ^ Bloodworth AJ, Bothwell BD, Collins AN, Maidwell NL. A short synthesis of naturally occurring and other analogues of plakinic acids that contain the 1,2-dioxolane group A short synthesis of naturally occurring and other analogues of plakinic acids that contain the 1,2-dioxolane group. Tetrahedron Letters. 1996, 37 (11): 1885–1888. doi:10.1016/0040-4039(96)00143-8.
- ^ 11.0 11.1 Phillipson DW, Rinehart Jr K. Antifungal peroxide-containing acids from two Caribbean sponges. Journal of the American Chemical Society. 1983, 105 (26): 7735–7736. doi:10.1021/ja00364a045.
- ^ Dai P, Trullinger TK, Liu X, Dussault PH. Asymmetric synthesis of 1,2-dioxolane-3-acetic acids: synthesis and configurational assignment of plakinic acid A. The Journal of Organic Chemistry. March 2006, 71 (6): 2283–92. PMID 16526775. doi:10.1021/jo0522254.
- ^ Kandur WV, Richert KJ, Rieder CJ, Thomas AM, Hu C, Ziller JW, Woerpel KA. Synthesis and reactivity of 1,2-dioxolanes from β,γ-epoxy ketones. Organic Letters. May 2014, 16 (10): 2650–3. PMID 24779430. doi:10.1021/ol500835f.
