戈尔德贝格反应
外观
(重定向自Goldberg反应)
戈尔德贝格反应(Goldberg reaction)是用苯胺或芳基酰胺[1][2][3]代替酚进行的Ullmann二芳醚合成。由德国女化学家、弗里茨·乌尔曼的妻子伊尔玛·戈尔德贝格[4]发现。

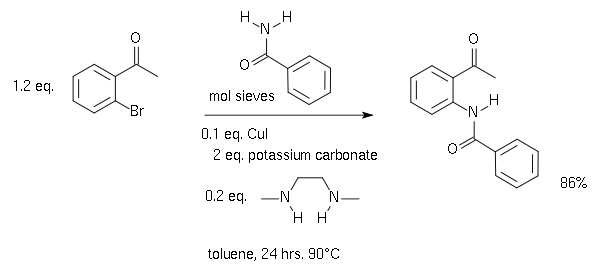
邻溴苯乙酮与苯甲酰胺进行偶联的产物,可以用作Camps环化反应的起始原料。[6]
戈尔德贝格等报道了一种用从碘化亚铜和邻二氮菲制备的催化剂介导的芳基碘化物和芳胺之间的乌尔曼型芳香胺化偶联反应。富电子的芳基碘化物也能进行反应,因此这种方法可用于弥补布赫瓦尔德-哈特维希反应有些情况下不适用于富电子芳卤的缺点。[7]
参见
[编辑]参考资料
[编辑]- ^ Klapars, A.; Antilla, J. C.; Huang, X.; Buchwald, S. L. A General and Efficient Copper Catalyst for the Amidation of Aryl Halides and the N-Arylation of Nitrogen Heterocycles. J. Am. Chem. Soc.(Communication). 2001, 123 (31): 7727–7729. doi:10.1021/ja016226z.
- ^ Klapars, A.; Huang, X.; Buchwald, S. L. A General and Efficient Copper Catalyst for the Amidation of Aryl Halides. J. Am. Chem. Soc.(Article). 2002, 124 (25): 7421–7428. doi:10.1021/ja0260465.
- ^ Strieter, E. R.; Blackmond, D. G.; Buchwald, S. L. The Role of Chelating Diamine Ligands in the Goldberg Reaction: A Kinetic Study on the Copper-Catalyzed Amidation of Aryl Iodides. J. Am. Chem. Soc.(Communication). 2005, 127 (12): 4120–4121. doi:10.1021/ja050120c.
- ^ Iram Goldberg. Ueber Phenylirungen bei Gegenwart von Kupfer als Katalysator. Berichte der deutschen chemischen Gesellschaft. 1906, 39 (2): 1691–1692. doi:10.1002/cber.19060390298.
- ^ C. F. H. Allen and G. H. W. McKee (1943). "Acridone". Org. Synth.; Coll. Vol. 2: 15.
- ^ Jones, C. P.; Anderson, K. W.; Buchwald, S. L. Sequential Cu-Catalyzed Amidation-Base-Mediated Camps Cyclization: A Two-Step Synthesis of 2-Aryl-4-quinolones from o-Halophenones. J. Org. Chem. 2007, 72 (21): 7968–7973. doi:10.1021/jo701384n.
- ^ H.B. Goodbrand, Nan-Xing Hu. Ligand-Accelerated Catalysis of the Ullmann Condensation: Application to Hole Conducting Triarylamines. Journal of Organic Chemistry. 1999, 64: 670–674. doi:10.1021/jo981804o.
