季節性流感疫苗
 美國海軍人員接種疫苗 | |
| 臨床資料 | |
|---|---|
| 商品名 | Fluarix、Fluzone、others |
| AHFS/Drugs.com | Monograph |
| 給藥途徑 | IM、鼻內 |
| ATC碼 | |
| 法律規範狀態 | |
| 法律規範 | |
| 識別資訊 | |
| CAS號 | 1704512-59-3 |
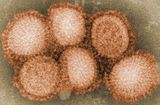
| 流行性感冒 |
|---|
 |
| 類型 |
| 疫苗 |
| 治療 |
| 大流行 |
| 爆發疫情 |
| 參見 |
季節性流感疫苗,簡稱流感疫苗(influenza vaccine,flu shot),為針對流行性感冒(季節性流感)的疫苗。因為流感病毒變化的速度很快,一年會發展新的流感疫苗兩次。大部分狀況下,疫苗有中度到高度的保護力;然而每年情況略有不同。[2][3]對65歲以上的長者,目前證據較缺乏。[4][5]平均來說,他們可以減少半日因病而上班必須請假的狀況。[6]有打疫苗的孩童,可以保護他們周遭的人。[2]
世界衛生組織和美國疾病控制與預防中心都建議幾乎任何大於六個月的人,每年應該施打流感疫苗。[2][7]尤其是孕婦、六個月大至五歲的孩童、有慢性疾病者、美洲原住民、以及醫療相關人員。[2][1]
分類
[編輯]流感疫苗多分為:(裂解)滅活疫苗(IIV)及裂解亞單位疫苗[8]、減毒活疫苗(LAIV)、重組亞單位疫苗(RIV)[9]。滅活疫苗(含滅活全病毒、滅活裂解、裂解亞單位三個子類[8])最為常用,又分為三價與四價劑型。
其中,對於流感的滅活疫苗是用滅活劑來處理在雞蛋或細胞培養物中製備的野生型病毒。如果季節性疫苗所包含的病毒在抗原上與最終流行的病毒非常匹配,滅活疫苗通常可以預防一半以上的嚴重流感相關的疾病[10]。然而,這些人群應禁止接種該疫苗[10]
- 接種流感疫苗後出現過危及生命的過敏反應
- 對疫苗中的任何成分(其中一些含有少量雞蛋蛋白)嚴重過敏的人
- 有格林-巴利綜合症病史的人
疫苗通常每年接種一劑,但 6 個月至 8 歲的兒童在一個流感季節可能需要接種兩劑。
第二種主要流感疫苗是通過鼻腔噴霧局部注射的三價或四價減毒活疫苗。每年都會製備新的疫苗,以解決抗原漂移問題[10]。該疫苗在美國許可用於 2 至 49 歲的人群[10]。
有效性
[編輯]過去滅活疫苗及減毒疫苗曾產生抗體依賴增強作用使2009年接種季節性甲型H1N1(針對類似A/Brisbane/59/2007的一株)疫苗的人更容易得甲型H1N1pdm09(A/California/7/2009病毒)新型流感。[11]2010年起H1N1株被改為「pandemic系」,2022年為人6B.1a.5a.2系。[12]2011年起開始推廣亞單位疫苗。
年度更新
[編輯]在每年的季節性流行期間,編碼血凝素和神經氨酸酶蛋白的基因都會發生點突變,而這兩種蛋白是保護性抗體的主要靶標。流行性流感病毒株的這種抗原漂移導致監管官員和生產商每年都要調整流感疫苗中的病毒抗原。偶爾,人類和禽類共同感染的情況下,分割的病毒基因組會重新出現,產生一種新的病毒,也就是抗原轉變,從而導致大流行。1918 年(H1N1)、1957 年(H2N2)、1968 年(H3N2)和 2009 年(新型 H1N1)都發生過全球性的大流行。在這種情況下,必須對疫苗進行重大調整[10]。
以下為北半球每年流感疫苗針對的毒株列表。Starting in the 2012–2013 season, the recommendation shifted from a trivalent influenza vaccine (TIV) containing three strains to a quadrivalent influenza vaccine (QIV) that contains both influenza B lineages.[來源請求]
| 年度 | H1N1甲流 | 演化支 | 類似豬H1支[note 1] | H3N2甲流 | 演化支 | 乙流Victoria株 | 演化支 | 乙流山形株 |
|---|---|---|---|---|---|---|---|---|
| November 1998–April 1999[13] | A/Beijing/262/95 (H1N1)-like virus | 未譜系 | A/Sydney/5/97 (H3N2)-like virus | N/A | B/Beijing/184/93-like virus | |||
| November 1999–April 2000[14] | B/Shangdong/7/97-like virus[note 2] | B/Beijing/184/93-like virus[note 2] | ||||||
| 2000–2001[15] | A/New Caledonia/20/99 (H1N1)-like virus | A/Moscow/10/99 (H3N2)-like virus | N/A | B/Beijing/184/93-like virus | ||||
| 2001–2002[16] | N/A | B/Sichuan/379/99-like virus | ||||||
| 2002–2003[17] | B/Hong Kong/330/2001-like virus | N/A | ||||||
| 2003–2004[18] | B/Hong Kong/330/2001-like virus | N/A | ||||||
| 2004–2005[19] | A/Fujian/411/2002 (H3N2)-like virus | N/A | B/Shanghai/361/2002-like virus | |||||
| 2005–2006[20] | A/California/7/2004 (H3N2)-like virus | N/A | B/Shanghai/361/2002-like virus | |||||
| 2006–2007[21] | A/Wisconsin/67/2005 (H3N2)-like virus | B/Malaysia/2506/2004-like virus | N/A | |||||
| 2007–2008[22] | A/Solomon Islands/3/2006 (H1N1)-like virus | B/Malaysia/2506/2004-like virus | N/A | |||||
| 2008–2009[23] | A/Brisbane/59/2007 (H1N1)-like virus | 1B.2.1[12] | A/Brisbane/10/2007 (H3N2)-like virus | N/A | B/Florida/4/2006-like virus | |||
| 2009–2010[24] | B/Brisbane/60/2008-like virus | N/A | ||||||
| 2010–2011[25] | A/California/7/2009 (H1N1)-like virus | (0)[26] | 1A.3.3.2[12] | A/Perth/16/2009 (H3N2)-like virus | N/A | |||
| 2011–2012[27] | A/California/7/2009 (H1N1)-like virus | N/A | ||||||
| 2012–2013[28] | A/California/7/2009 (H1N1)pdm09[note 3]-like virus[29] | A/Victoria/361/2011 (H3N2)-like virus | B/Wisconsin/1/2010-like virus | |||||
| 2013–2014[30] | A/California/7/2009 (H1N1)pdm09[note 3]-like virus | A(H3N2) virus antigenically like the cell-propagated prototype virus A/Victoria/361/2011[note 4] | B/Massachusetts/2/2012-like virus | |||||
| 2014–2015[31] | A/California/7/2009 (H1N1)pdm09[note 3]-like virus | A/Texas/50/2012 (H3N2)-like virus[note 5] | ||||||
| 2015–2016[32] | A/California/7/2009 (H1N1)pdm09[note 3]-like virus | A/Switzerland/9715293/2013 (H3N2)-like virus | B/Phuket/3073/2013-like virus | |||||
| 2016–2017[33][34] | A/California/7/2009 (H1N1)pdm09[note 3]-like virus | A/Hong Kong/4801/2014 (H3N2)-like virus | 3C.2a | |||||
| 2017–2018[35][36] | A/Michigan/45/2015 (H1N1)pdm09[note 3]-like virus[37] | 6B.1 | 不再使用 | |||||
| 2018–2019[38][39] | A/Michigan/45/2015 (H1N1)pdm09[note 3]-like virus | A/Singapore/INFIMH-16-0019/2016 (H3N2)-like virus | 3C.2a1 | B/Colorado/06/2017-like virus (B/Victoria/2/87 lineage) | ||||
| 2019–2020[40][41][42] | A/Brisbane/02/2018 (H1N1)pdm09[note 3]-like virus | 6B.1A.1 | A/Kansas/14/2017 (H3N2)-like virus | 3C.3a | ||||
| 2020–2021 egg-based vaccines[43] | A/Guangdong-Maonan/SWL1536/2019 (H1N1)pdm09-like virus[note 3] | 6B.1A.5a.1 | A/Hong Kong/2671/2019 (H3N2)-like virus | 3C.2a1b.1b | B/Washington/02/2019 (B/Victoria lineage)-like virus | V1A.3 | ||
| 2020–2021 cell- or recombinant-based vaccines[43] | A/Hawaii/70/2019 (H1N1)pdm09-like virus[note 3] | A/Hong Kong/45/2019 (H3N2)-like virus | ||||||
| 2021–2022 egg-based vaccines[44] | A/Victoria/2570/2019 (H1N1)pdm09-like virus[note 3] | 6B.1A.5a.2 | A/Cambodia/e0826360/2020 (H3N2)-like virus | 3C.2a1b.2a.1 | ||||
| 2021–2022 cell- or recombinant-based vaccines[44] | A/Wisconsin/588/2019 (H1N1)pdm09-like virus[note 3] | |||||||
| 2022–2023 egg-based vaccines[45] | A/Victoria/2570/2019 (H1N1)pdm09-like virus | A/Darwin/9/2021 (H3N2)-like virus | 3C.2a1b.2a.2a | B/Austria/1359417/2021 (B/Victoria lineage)-like virus | V1A.3a.2 | |||
| 2022–2023 cell- or recombinant-based vaccines[45] | A/Wisconsin/588/2019 (H1N1)pdm09-like virus | A/Darwin/6/2021 (H3N2)-like virus | ||||||
| 2023–2024 egg-based vaccines[46] | A/Victoria/4897/2022 (H1N1)pdm09-like virus | 6B.1A.5a.2a.1 | A/Darwin/9/2021 (H3N2)-like virus | |||||
| 2023–2024 cell- or recombinant-based vaccines[46] | A/Wisconsin/67/2022 (H1N1)pdm09-like virus | A/Darwin/6/2021 (H3N2)-like virus |
- ^ 早期針對人類流感病毒進行詳細譜系,僅存在最接近的豬H1流感病毒的HA蛋白譜系,一些最接近人H1N1流感病毒的豬H1流感病毒甚至屬於H1N2型。
- ^ 2.0 2.1 The recommendation was for either the B Victoria or the B Yamagata strain.
- ^ 3.00 3.01 3.02 3.03 3.04 3.05 3.06 3.07 3.08 3.09 3.10 3.11 (H1N1)pdm09 is newer nomenclature for the 2009 pandemic H1N1 virus, not a different strain.
- ^ It is recommended that A/Texas/50/2012 is used as the A(H3N2) vaccine component because of antigenic changes in earlier A/Victoria/361/2011-like vaccine viruses (such as IVR-165) resulting from adaptation to propagation in eggs.
- ^ A/Texas/50/2012 is an A(H3N2) virus that following adaptation to growth in eggs has maintained antigenic properties similar to the majority of recently circulating cell-propagated A(H3N2) viruses including A/Victoria/361/2011
副作用
[編輯]疫苗大致上是安全的。在兒童,有5-10%會有發燒的反應,也有可能有肌肉痛、疲倦等症狀。在某些年份,疫苗可能在年長者引起格林-巴利綜合症,概率約百萬分之一。疫苗不應該施打於對蛋嚴重過敏者、或有流感疫苗過敏史者。疫苗有滅活疫苗及減毒疫苗兩種。孕婦應該使用滅活疫苗。在施打類型方面,有肌肉內注射、鼻噴劑、皮內注射等形式。[2]
歷史
[編輯]對抗流感的疫苗在1930年代開始,一直到1945年開始在美國廣為使用。[47][48]它被列在世界衛生組織基本藥物標準清單,即其建議之對基礎健康照護系統最重要的藥物。[49]在2014年,其批發價約5.25美元。[50]在美國,則是少於25美元。[51]
參考
[編輯]- ^ 1.0 1.1 Influenza Virus Vaccine Inactivated. The American Society of Health-System Pharmacists. [2015-01-08]. (原始內容存檔於2014-12-25).
- ^ 2.0 2.1 2.2 2.3 2.4 Vaccines against influenza WHO position paper – November 2012. Releve Epidemiologique Hebdomadaire. 2012-11-23, 87 (47) [2022-11-02]. ISSN 0049-8114. PMID 23210147. (原始內容存檔於2022-11-02).
- ^ Manzoli, Lamberto; Ioannidis, John P.A.; Flacco, Maria Elena; De Vito, Corrado; Villari, Paolo. Effectiveness and harms of seasonal and pandemic influenza vaccines in children, adults and elderly: A critical review and re-analysis of 15 meta-analyses. Human Vaccines & Immunotherapeutics. 2012-07, 8 (7) [2022-11-02]. ISSN 2164-5515. PMC 3495721
 . PMID 22777099. doi:10.4161/hv.19917. (原始內容存檔於2020-06-07) (英語).
. PMID 22777099. doi:10.4161/hv.19917. (原始內容存檔於2020-06-07) (英語).
- ^ Osterholm, Michael T.; Kelley, Nicholas S.; Sommer, Alfred; Belongia, Edward A. Efficacy and effectiveness of influenza vaccines: a systematic review and meta-analysis. The Lancet Infectious Diseases. 2012-01-01, 12 (1) [2022-11-02]. ISSN 1473-3099. PMID 22032844. doi:10.1016/S1473-3099(11)70295-X. (原始內容存檔於2018-09-14) (英語).
- ^ Jefferson, Tom; Di Pietrantonj, Carlo; Al-Ansary, Lubna A.; Ferroni, Eliana; Thorning, Sarah; Thomas, Roger E. Vaccines for preventing influenza in the elderly. The Cochrane Database of Systematic Reviews. 2010-02-17, (2) [2022-11-02]. ISSN 1469-493X. PMID 20166072. doi:10.1002/14651858.CD004876.pub3. (原始內容存檔於2022-11-23).
- ^ Demicheli, Vittorio; Jefferson, Tom; Al-Ansary, Lubna A.; Ferroni, Eliana; Rivetti, Alessandro; Di Pietrantonj, Carlo. Vaccines for preventing influenza in healthy adults. The Cochrane Database of Systematic Reviews. 2014-03-13, (3) [2022-11-02]. ISSN 1469-493X. PMID 24623315. doi:10.1002/14651858.CD001269.pub5. (原始內容存檔於2023-03-15).
- ^ Who Should Get Vaccinated Against Influenza. U.S. Centers for Disease Control and Prevention. 2015-11-04 [2015-12-08]. (原始內容存檔於2014-01-22).
- ^ 8.0 8.1 Sook-San Wong and Richard J. Webby. Traditional and New Influenza Vaccines. Clin Microbiol Rev. 2013 Jul; 26(3): 476–492. [2023-03-14]. doi:10.1128/CMR.00097-12. (原始內容存檔於2023-03-14).
- ^ Comparison of live attenuated influenza vaccine (LAIV), inactivated influenza vaccine (IIV), and recombinant influenza vaccine (RIV) for prevention of seasonal influenza, United States formulations for persons younger than 65 years. [2022-11-03]. (原始內容存檔於2022-11-03).
- ^ 10.0 10.1 10.2 10.3 10.4 Brunton, Laurence L; Hilal-Dandan, Randa; Knollmann, Björn C. Goodman Gilmans The Pharmacological Basis of Therapeutics. The United States: McGraw-Hill Education. 2018: 666–667. ISBN 978-1-25-958474-9.
- ^ Skowronski DM, De Serres G, Crowcroft NS, Janjua NZ, Boulianne N, Hottes TS, et al. Association between the 2008-09 seasonal influenza vaccine and pandemic H1N1 illness during Spring-Summer 2009: four observational studies from Canada. PLOS Medicine. April 2010, 7 (4): e1000258. PMC 2850386
 . PMID 20386731. doi:10.1371/journal.pmed.1000258.
. PMID 20386731. doi:10.1371/journal.pmed.1000258.
- ^ 12.0 12.1 12.2 Tapia, R.; Torremorell, M.; Culhane, M.; et al. Antigenic characterization of novel H1 influenza A viruses in swine. [2023-03-14]. (原始內容存檔於2023-03-16).
- ^ Recommendations for Influenza Vaccine Composition Northern hemisphere November 1998-April 1999. World Health Organization (WHO). [14 June 2012]. (原始內容存檔於29 April 2013).
- ^ Recommendations for Influenza Vaccine Composition Northern hemisphere: November 1999 to April 2000. World Health Organization (WHO). [14 June 2012]. (原始內容存檔於29 April 2013).
- ^ Recommendations for Influenza Vaccine Composition Northern hemisphere: 2000-2001. World Health Organization (WHO). [14 June 2012]. (原始內容存檔於29 April 2013).
- ^ Recommendations for Influenza Vaccine Composition Northern hemisphere: 2001-2002. World Health Organization (WHO). [14 June 2012]. (原始內容存檔於29 April 2013).
- ^ Recommendations for Influenza Vaccine Composition Northern hemisphere: 2002-2003. World Health Organization (WHO). [14 June 2012]. (原始內容存檔於29 April 2013).
- ^ Recommendations for Influenza Vaccine Composition Northern hemisphere: 2003-2004. World Health Organization (WHO). [14 June 2012]. (原始內容存檔於29 April 2013).
- ^ Recommendations for Influenza Vaccine Composition Northern hemisphere: 2004-2005. World Health Organization (WHO). [14 June 2012]. (原始內容存檔於29 April 2013).
- ^ Recommendations for Influenza Vaccine Composition Northern hemisphere: 2005-2006. World Health Organization (WHO). [14 June 2012]. (原始內容存檔於29 April 2013).
- ^ Recommendations for Influenza Vaccine Composition Northern hemisphere: 2006-2007. World Health Organization (WHO). [14 June 2012]. (原始內容存檔於29 April 2013).
- ^ Recommendations for Influenza Vaccine Composition Northern hemisphere: 2007-2008. World Health Organization (WHO). [14 June 2012]. (原始內容存檔於29 April 2013).
- ^ Recommendations for Influenza Vaccine Composition Northern hemisphere: 2008-2009. World Health Organization (WHO). [14 June 2012]. (原始內容存檔於29 April 2013).
- ^ Recommendations for Influenza Vaccine Composition Northern hemisphere: 2009-2010. World Health Organization (WHO). [14 June 2012]. (原始內容存檔於29 April 2013).
- ^ Recommended viruses for influenza vaccines for use in the 2010-2011 northern hemisphere influenza season. World Health Organization (WHO). [14 June 2012]. (原始內容存檔於28 October 2013).
- ^ 不屬於1~7,為1~7系的祖型
- ^ Recommended composition of influenza virus vaccines for use in the 2011-2012 northern hemisphere influenza season. World Health Organization (WHO). [14 June 2012]. (原始內容存檔於9 October 2011).
- ^ Recommended composition of influenza virus vaccines for use in the 2012-2013 northern hemisphere influenza season. World Health Organization (WHO). [14 June 2012]. (原始內容存檔於27 February 2012).
- ^ Standardization of terminology of the pandemic A(H1N1) 2009 virus (PDF). World Health Organization (WHO). [14 June 2012]. (原始內容存檔 (PDF)於2022-03-08).
- ^ Recommended composition of influenza virus vaccines for use in the 2013-2014 northern hemisphere influenza season. World Health Organization (WHO). [22 February 2013]. (原始內容存檔於1 March 2013).
- ^ Recommended composition of influenza virus vaccines for use in the 2014-2015 northern hemisphere influenza season. World Health Organization (WHO). [20 February 2014]. (原始內容存檔於25 February 2014).
- ^ Recommended composition of influenza virus vaccines for use in the 2015-2016 northern hemisphere influenza season. World Health Organization (WHO). [2 March 2015]. (原始內容存檔於6 March 2015).
- ^ Recommended composition of influenza virus vaccines for use in the 2016-2017 northern hemisphere influenza season. World Health Organization (WHO). [19 May 2016]. (原始內容存檔於4 March 2016).
- ^ World Health Organization. Recommended composition of influenza virus vaccines for use in the 2016–2017 northern hemisphere influenza season. Weekly Epidemiological Record. March 2016, 91 (10): 121–32. PMID 26971356. hdl:10665/254302.
- ^ Recommended composition of influenza virus vaccines for use in the 2017-2018 northern hemisphere influenza season. World Health Organization (WHO). [16 March 2017]. (原始內容存檔於7 March 2017).
- ^ World Health Organization. Recommended composition of influenza virus vaccines for use in the 2017–2018 northern hemisphere influenza season. Weekly Epidemiological Record. March 2017, 92 (11): 117–28. PMID 28303704. hdl:10665/254758.
- ^ WHO switches H1N1 in Southern Hemisphere flu vaccine. University of Minnesota - Center for Infectious Disease Research and Policy (CIDRAP). 29 September 2016 [12 September 2017]. (原始內容存檔於2017-09-12).
A/Michigan/45/2015 replaces the longstanding A/California/7/2009 and is recommended to improve protection against two subclades that have emerged over the past season.
- ^ Recommended composition of influenza virus vaccines for use in the 2018-2019 northern hemisphere influenza season. World Health Organization (WHO). [22 February 2018]. (原始內容存檔於26 February 2018).
- ^ World Health Organization. Recommended composition of influenza virus vaccines for use in the 2018–2019 northern hemisphere influenza season. Weekly Epidemiological Record. March 2018, 93 (12): 133–41. PMID 29569429. hdl:10665/272270.
- ^ Recommended composition of influenza virus vaccines for use in the 2019-2020 northern hemisphere influenza season. World Health Organization (WHO). [22 March 2019]. (原始內容存檔於29 March 2022).
- ^ World Health Organization. Recommended composition of influenza virus vaccines for use in the 2019–2020 northern hemisphere influenza season. Weekly Epidemiological Record. March 2019, 94 (12): 141–50. hdl:10665/311441.
- ^ World Health Organization. Addendum to the Recommended composition of influenza virus vaccines for use in the 2019–2020 northern hemisphere influenza season. Weekly Epidemiological Record. March 2019, 94 (13): 166–8. hdl:10665/311555.
- ^ 43.0 43.1 Recommended composition of influenza virus vaccines for use in the 2020-2021 northern hemisphere influenza season. World Health Organization (WHO). [28 February 2020]. (原始內容存檔於12 March 2020).
- ^ 44.0 44.1 Recommended composition of influenza virus vaccines for use in the 2021-2022 northern hemisphere influenza season. World Health Organization (WHO). [26 February 2021]. (原始內容存檔於26 February 2021).
- ^ 45.0 45.1 Recommended composition of influenza virus vaccines for use in the 2022-2023 northern hemisphere influenza season. World Health Organization (WHO). 25 February 2022 [29 March 2022]. (原始內容存檔於29 March 2022).
- ^ 46.0 46.1 Recommended composition of influenza virus vaccines for use in the 2023-2024 northern hemisphere influenza season. World Health Organization (WHO). 24 February 2023 [17 March 2023]. (原始內容存檔於8 March 2023).
- ^ Compans, Richard W.; Orenstein, Walter A. Vaccines for Pandemic Influenza. Springer Science & Business Media. 2009-09-18: 49 [2016-03-12]. ISBN 978-3-540-92165-3. (原始內容存檔於2022-10-24) (英語).
- ^ Nunnally, Brian K.; Turula, Vincent E.; Sitrin, Robert D. Vaccine Analysis: Strategies, Principles, and Control. Springer. 2014-11-27: 61 [2016-03-12]. ISBN 978-3-662-45024-6. (原始內容存檔於2022-10-24) (英語).
- ^ 19th WHO Model List of Essential Medicines (April 2015) (PDF). WHO. April 2015 [2015-05-10]. (原始內容存檔 (PDF)於2015-05-13).
- ^ Vaccine, influenza. International Drug Price Indicator Guide. [2015-12-06].[失效連結]
- ^ Hamilton, Richart. Tarascon Pocket Pharmacopoeia 2015 Deluxe Lab-Coat Edition. Jones & Bartlett Learning. 2015: 314. ISBN 9781284057560.
