腺相关病毒
| 腺相关病毒 | |
|---|---|
| |
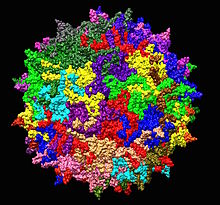
| 一种用作人基因治疗载体的腺相关病毒1LP3的3D原子结构 | |
| 病毒分类 | |
| (未分级): | 病毒 Virus |
| 域: | 单链DNA病毒域 Monodnaviria |
| 界: | 称德病毒界 Shotokuvirae |
| 门: | 科萨特病毒门 Cossaviricota |
| 纲: | 第五病毒纲 Quintoviricetes |
| 目: | 小病毒目 Piccovirales |
| 科: | 细小病毒科 Parvoviridae |
| 属: | 依赖性细小病毒属 Dependoparvovirus |
| 种: | 腺相关病毒 Adeno-associated virus
|
| 种类 | |
| |
腺相关病毒(Adeno-associated viruses,常缩写为AAV),是一种隶属细小病毒科依赖性细小病毒属、能够感染人类以及其他部分灵长类动物的病毒。腺相关病毒直径约20纳米、无被膜,无法自主完成复制,全基因组长约4.8千碱基对(kb)[1][2]。
尚无已知的疾病与腺相关病毒有关。腺相关病毒一般只能引发轻度的免疫反应。加之腺相关病毒须在其他病毒同时感染宿主的前提下才能复制,使研究者认为腺相关病毒适合用于改造用作人基因治疗的载体,以及体外等基因人类疾病模型的构建[3]。由腺病毒改造而成的基因治疗载体能够同时感染分裂中和分裂不活跃的细胞,并将载体DNA以一种不整合入染色体的方式导入到细胞中(不过在自然条件下,也有腺相关病毒携带的DNA在感染后插入染色体的报导)[4]。目前,一部分使用腺相关病毒的基因治疗临床试验已取得正面的结果[5]。
研究历史
[编辑]腺相关病毒最初被认为是腺病毒制备过程中混入的污染物之一。20世纪60年代,匹兹堡大学的鲍勃·艾奇逊(Bob Atchison)与美国国立卫生研究院的华莱士·罗(Wallace P. Rowe)实验室的工作最初确定腺相关病毒是一种依赖性细小病毒。后来的血清学研究表明,腺相关病毒不能造成任何已知的人类疾病,且只能在腺病毒、疱疹病毒等辅助病毒存在的前提下才能感染人类[6]。
复制周期
[编辑]
绝大部分情况下,腺相关病毒需要辅助病毒的存在才能完成完整的生命周期[7]。腺相关病毒在感染细胞后,需要辅助病毒的帮助(例如,腺相关病毒的辅助病毒疱疹病毒能为其提供DNA聚合酶和解旋酶以及一些对腺相关病毒转录的早期启动必要的蛋白)才能进入裂解期,进行病毒复制。在没有辅助病毒的前提下,腺相关病毒基因的表达将会受限,一部分腺相关病毒DNA会在这种情况下插入至人19号染色体q13.4区域,即AAVS1位点[8]。
基因组
[编辑]
腺相关病毒的基因组由一条长约4.8千碱基对(kb)的正义或反义单链DNA构成,其两端是序列对称的反向重复序列(ITR),对腺相关病毒的复制[9]、衣壳化[10]、以及插入宿主染色体的过程有重要作用[11][12] ,中间则有rep与cap两个开放读框,前者包括四个序列相互重叠的基因,编码Rep78、Rep68、Rep52与Rep40等四种参与病毒基因复制的蛋白;后者包括三个序列重叠的基因,编码VP1、VP2与VP3等三种组成病毒衣壳的蛋白[13]。腺相关病毒本身不编码DNA聚合酶,仅仰赖宿主细胞或辅助病毒的DNA聚合酶合成自己的基因组[14]。
反向重复序列
[编辑]腺相关病毒基因组两端的反向重复序列(ITR)序列对称,长145碱基,可各自形成一茎环结构,使病毒利用其3'活性端开始DNA复制,无需由引物酶合成引物即可复制自身的DNA[14],形成相互连结的双股DNA中间产物,再以缺口酶将两股DNA切开[14]。除DNA复制外,反向重复序列也参与病毒插入宿主DNA的过程[11][12],且为病毒颗粒正常组装所需[10]。
rep基因
[编辑]腺相关病毒基因组中上游的rep读框可由左侧的p5或p19两个启动子开始转录,产生长度不一的两种RNA,这两种RNA中皆含有一个内含子,可能在转录后被切除,依使用启动子的不同与内含子被切除与否,rep基因可转录出四种mRNA,进而产生四种蛋白质变体:Rep78、Rep68、Rep52,以及Rep40(Rep后面的数字代表这种蛋白的分子量为多少千道尔顿)[15],前两者是由p5启动子转录产生,可与反向重复序列形成的茎环结合,参与病毒DNA的复制,后两者则是由p19启动子转录产生[16]。四种Rep蛋白皆可与ATP结合,且皆具解旋酶活性,它们可抑制自身的启动子(p5和p19)转录,并可激活转录cap基因蛋白的p40启动子[15][17][18][19][20]。
cap基因
[编辑]cap基因位于rep基因的下游,p40启动子可调控其转录,产生序列重叠的VP1、VP2、VP3与AAP(组装激活蛋白)四种蛋白,VP1、VP2与VP3的大小分别为87、72与62千道尔顿[21],可以1:1:10的比例组成腺相关病毒二十面体的衣壳[22],AAP亦为衣壳组装所需的蛋白[23]。cap基因转录出的RNA可能将一段较长的内含子或一段较短的内含子切除,分别形成长2.3kb或长2.6kb的mRNA,其中长内含子被切除的情况较为常见,因此2.3kb的mRNA为大宗,可翻译产生VP3蛋白,但其起始密码子(AUG)上游有一个ACG密码子周边亦形成可起始翻译的Kozak序列,少数核糖体会自此开始翻译,产生比VP3蛋白多出一N端延伸区域的VP2蛋白;而切除短内含子的2.6kb的mRNA则会翻译产生VP1蛋白,也比VP3多了一N端延伸区[24][25][26][27]。
由于2.3mRNA的数量较多,且其上游ACG密码子启动翻译的能力比正常的起始密码子弱,三种衣壳蛋白中VP3被翻译生成的量远多于VP1与VP2,与其在衣壳中的组成比例相符[28]。有研究显示VP1蛋白的N端延伸区有磷脂酶A2活性,可能有助于病毒自细胞释出[29],因而为病毒复制周期所需,相较之下VP2可能并非病毒组装必须的蛋白[30]。
分类
[编辑]腺相关病毒在人类与其他灵长类中相当普遍,目前已知有11个血清型[31],其中AAV-2、AAV-3、AAV-5与AAV-6为自人类细胞发现,AAV-1、AAV-4与AAV-7至AAV-11为自其他灵长类细胞发现[32],其中对AAV-2的研究最多[33][34][35],此血清型可能使用硫酸肝素蛋白多糖(HSPG)、aVβ5整合素与成纤维细胞生长因子受体1(FGFR-1)为受体感染细胞,其中前者为主要受体,后两者为辅受体,可协助病毒以底物媒介内吞作用进入细胞[36][37][38],但学界对此尚有争议[39]。
2013年起,国际病毒分类委员会(ICTV)将腺相关病毒分为A型与B型两个物种,前者包含AAV-1、AAV-2、AAV-3与AAV-4,后者即AAV-5[40][41]。
为提升在腺相关病毒在临床与研究中作为载体的效率,研究人员研发了许多腺相关病毒的人工变型,以提升基因表达、适应不同的目标组织并使其更易逃避免疫系统的攻击,例如AAV-2的基因组与AAV-5的衣壳蛋白基因融合组成的AAV-2/5,以及由8型AAV序列融合组成的AAV-DJ 等[42]。
用于基因治疗
[编辑]腺相关病毒因毒性低、不能自主复制、不致病,且较少整合到宿主基因组中,因而被认为是一种较为理想的基因治疗载体。将腺相关病毒改造为基因治疗载体的具体操作方法是腺相关病毒基因组上的rep与cap区域替换为目的基因[3][43]。腺相关病毒载体导入人体后,不分裂的细胞能够一直保持输入的目的基因片段,而持续分裂细胞则会逐渐随细胞分裂丢失经由腺相关病毒导入的基因片段,因此腺相关病毒疗法不太适合以持续分裂的细胞(如干细胞)作为靶标[43]。
腺相关病毒作为基因治疗载体存在一些缺点。首先是容量低,最多只能容纳4.5kb的插入片段,无法容纳DMD等较长的人类基因[43]。其次,输入的腺相关病毒载体可能被体内存在的针对天然腺相关病毒的抗体中和。此外,腺相关病毒也存在整合入宿主基因组的情况[44]。
过去认为将腺相关病毒用于基因治疗时,反向重复序列是病毒基因组中唯一需与转入的基因顺式(in cis)组装的元件,cap与rep蛋白皆可反式(in trans)导入,但有新研究结果显示rep基因编码区域中有一称为顺式rep依赖型元件(cis-acting Rep-dependent element,简称CARE)的序列与转入的基因顺式组装时也可促进病毒的复制与组装[45][46][47][48]。
根据2019年的数据,全球范围内共有超过250件使用腺相关病毒载体技术的临床试验,占基于病毒载体的基因治疗临床试验总数的8.3%[49]。
参见
[编辑]参考文献
[编辑]- ^ Naso, Michael F.; Tomkowicz, Brian; Perry, William L.; Strohl, William R. Adeno-Associated Virus (AAV) as a Vector for Gene Therapy. BioDrugs. 2017, 31 (4): 317–334. ISSN 1173-8804. doi:10.1007/s40259-017-0234-5.
- ^ Wu, Zhijian; Yang, Hongyan; Colosi, Peter. Effect of Genome Size on AAV Vector Packaging. Molecular Therapy. 2010, 18 (1): 80–86. ISSN 1525-0016. doi:10.1038/mt.2009.255.
- ^ 3.0 3.1 Grieger JC, Samulski RJ. Adeno-associated Virus as a Gene Therapy Vector: Vector Development, Production and Clinical Applications. Adeno-associated virus as a gene therapy vector: vector development, production and clinical applications. Advances in Biochemical Engineering/Biotechnology 99. 2005: 119–45. ISBN 978-3-540-28404-8. PMID 16568890. doi:10.1007/10_005.
- ^ Deyle DR, Russell DW. Adeno-associated virus vector integration. Current Opinion in Molecular Therapeutics. August 2009, 11 (4): 442–7. PMC 2929125
 . PMID 19649989.
. PMID 19649989.
- ^ Maguire AM, Simonelli F, Pierce EA, Pugh EN, Mingozzi F, Bennicelli J, et al. Safety and efficacy of gene transfer for Leber's congenital amaurosis. The New England Journal of Medicine. May 2008, 358 (21): 2240–8. PMC 2829748
 . PMID 18441370. doi:10.1056/NEJMoa0802315.
. PMID 18441370. doi:10.1056/NEJMoa0802315.
- ^ Carter BJ. Adeno-associated virus and the development of adeno-associated virus vectors: a historical perspective. Molecular Therapy. 2004, 10 (6): 981–9. PMID 15564130. doi:10.1016/j.ymthe.2004.09.011
 .
.
- ^ Adeno-Associated Virus and Adeno-associated Viral Vectors. [2018-09-19]. (原始内容存档于2018-09-20).
- ^ Daya, Shyam; Berns, Kenneth I. Gene Therapy Using Adeno-Associated Virus Vectors. Clinical Microbiology Reviews. 2008, 21 (4): 583–593. ISSN 0893-8512. doi:10.1128/CMR.00008-08.
- ^ Bohenzky RA, LeFebvre RB, Berns KI. Sequence and symmetry requirements within the internal palindromic sequences of the adeno-associated virus terminal repeat. Virology. October 1988, 166 (2): 316–27. PMID 2845646. doi:10.1016/0042-6822(88)90502-8.
- ^ 10.0 10.1 Zhou X, Muzyczka N. In vitro packaging of adeno-associated virus DNA. Journal of Virology. April 1998, 72 (4): 3241–7. PMC 109794
 . PMID 9525651. doi:10.1128/JVI.72.4.3241-3247.1998.
. PMID 9525651. doi:10.1128/JVI.72.4.3241-3247.1998.
- ^ 11.0 11.1 Wang XS, Ponnazhagan S, Srivastava A. Rescue and replication signals of the adeno-associated virus 2 genome. Journal of Molecular Biology. July 1995, 250 (5): 573–80. PMID 7623375. doi:10.1006/jmbi.1995.0398.
- ^ 12.0 12.1 Weitzman MD, Kyöstiö SR, Kotin RM, Owens RA. Adeno-associated virus (AAV) Rep proteins mediate complex formation between AAV DNA and its integration site in human DNA. Proceedings of the National Academy of Sciences of the United States of America. June 1994, 91 (13): 5808–12. Bibcode:1994PNAS...91.5808W. PMC 44086
 . PMID 8016070. doi:10.1073/pnas.91.13.5808.
. PMID 8016070. doi:10.1073/pnas.91.13.5808.
- ^ Carter BJ. Adeno-associated virus and adeno-associated virus vectors for gene delivery. Lassic DD, Templeton NS (编). Gene Therapy: Therapeutic Mechanisms and Strategies. New York City: Marcel Dekker, Inc. 2000: 41–59. ISBN 978-0-585-39515-9.
- ^ 14.0 14.1 14.2 Gonçalves MA. Adeno-associated virus: from defective virus to effective vector.. Virol J. 2005, 2: 43. PMC 1131931
 . PMID 15877812. doi:10.1186/1743-422X-2-43.
. PMID 15877812. doi:10.1186/1743-422X-2-43.
- ^ 15.0 15.1 Kyöstiö SR, Owens RA, Weitzman MD, Antoni BA, Chejanovsky N, Carter BJ. Analysis of adeno-associated virus (AAV) wild-type and mutant Rep proteins for their abilities to negatively regulate AAV p5 and p19 mRNA levels. Journal of Virology. May 1994, 68 (5): 2947–57. PMC 236783
 . PMID 8151765. doi:10.1128/JVI.68.5.2947-2957.1994.
. PMID 8151765. doi:10.1128/JVI.68.5.2947-2957.1994.
- ^ Sitaraman V, Hearing P, Ward CB, Gnatenko DV, Wimmer E, Mueller S; et al. Computationally designed adeno-associated virus (AAV) Rep 78 is efficiently maintained within an adenovirus vector.. Proc Natl Acad Sci U S A. 2011, 108 (34): 14294–9. PMC 3161519
 . PMID 21844368. doi:10.1073/pnas.1102883108.
. PMID 21844368. doi:10.1073/pnas.1102883108.
- ^ Im DS, Muzyczka N. The AAV origin binding protein Rep68 is an ATP-dependent site-specific endonuclease with DNA helicase activity. Cell. May 1990, 61 (3): 447–57. PMID 2159383. S2CID 27997617. doi:10.1016/0092-8674(90)90526-K.
- ^ Im DS, Muzyczka N. Partial purification of adeno-associated virus Rep78, Rep52, and Rep40 and their biochemical characterization. Journal of Virology. February 1992, 66 (2): 1119–28. PMC 240816
 . PMID 1309894. doi:10.1128/JVI.66.2.1119-1128.1992.
. PMID 1309894. doi:10.1128/JVI.66.2.1119-1128.1992.
- ^ Samulski RJ. AAV vectors, the future workhorse of human gene therapy. Human Gene Therapy: Current Opportunities and Future Trends. 2003: 25–40. ISBN 978-3-662-05354-6. PMID 12894449. doi:10.1007/978-3-662-05352-2_3.
|journal=被忽略 (帮助);|issue=被忽略 (帮助) - ^ Trempe JP, Carter BJ. Regulation of adeno-associated virus gene expression in 293 cells: control of mRNA abundance and translation. Journal of Virology. January 1988, 62 (1): 68–74. PMC 250502
 . PMID 2824856. doi:10.1128/JVI.62.1.68-74.1988.
. PMID 2824856. doi:10.1128/JVI.62.1.68-74.1988.
- ^ Jay FT, Laughlin CA, Carter BJ. Eukaryotic translational control: adeno-associated virus protein synthesis is affected by a mutation in the adenovirus DNA-binding protein. Proceedings of the National Academy of Sciences of the United States of America. May 1981, 78 (5): 2927–31. Bibcode:1981PNAS...78.2927J. PMC 319472
 . PMID 6265925. doi:10.1073/pnas.78.5.2927.
. PMID 6265925. doi:10.1073/pnas.78.5.2927.
- ^ Sonntag F, Schmidt K, Kleinschmidt JA. A viral assembly factor promotes AAV2 capsid formation in the nucleolus. Proceedings of the National Academy of Sciences of the United States of America. June 2010, 107 (22): 10220–5. Bibcode:2010PNAS..10710220S. PMC 2890453
 . PMID 20479244. doi:10.1073/pnas.1001673107.
. PMID 20479244. doi:10.1073/pnas.1001673107.
- ^ Sonntag F, Köther K, Schmidt K, Weghofer M, Raupp C, Nieto K, Kuck A, Gerlach B, Böttcher B, Müller OJ, Lux K, Hörer M, Kleinschmidt JA. The assembly-activating protein promotes capsid assembly of different adeno-associated virus serotypes. Journal of Virology. December 2011, 85 (23): 12686–97. PMC 3209379
 . PMID 21917944. doi:10.1128/JVI.05359-11.
. PMID 21917944. doi:10.1128/JVI.05359-11.
- ^ Becerra SP, Rose JA, Hardy M, Baroudy BM, Anderson CW. Direct mapping of adeno-associated virus capsid proteins B and C: a possible ACG initiation codon. Proceedings of the National Academy of Sciences of the United States of America. December 1985, 82 (23): 7919–23. Bibcode:1985PNAS...82.7919B. PMC 390881
 . PMID 2999784. doi:10.1073/pnas.82.23.7919.
. PMID 2999784. doi:10.1073/pnas.82.23.7919.
- ^ Cassinotti P, Weitz M, Tratschin JD. Organization of the adeno-associated virus (AAV) capsid gene: mapping of a minor spliced mRNA coding for virus capsid protein 1. Virology. November 1988, 167 (1): 176–84. PMID 2847413. doi:10.1016/0042-6822(88)90067-0.
- ^ Muralidhar S, Becerra SP, Rose JA. Site-directed mutagenesis of adeno-associated virus type 2 structural protein initiation codons: effects on regulation of synthesis and biological activity. Journal of Virology. January 1994, 68 (1): 170–6. PMC 236275
 . PMID 8254726. doi:10.1128/JVI.68.1.170-176.1994.
. PMID 8254726. doi:10.1128/JVI.68.1.170-176.1994.
- ^ Trempe JP, Carter BJ. Alternate mRNA splicing is required for synthesis of adeno-associated virus VP1 capsid protein. Journal of Virology. September 1988, 62 (9): 3356–63. PMC 253458
 . PMID 2841488. doi:10.1128/JVI.62.9.3356-3363.1988.
. PMID 2841488. doi:10.1128/JVI.62.9.3356-3363.1988.
- ^ Rabinowitz JE, Samulski RJ. Building a better vector: the manipulation of AAV virions. Virology. December 2000, 278 (2): 301–8. PMID 11118354. doi:10.1006/viro.2000.0707
 .
.
- ^ Girod A, Wobus CE, Zádori Z, Ried M, Leike K, Tijssen P, Kleinschmidt JA, Hallek M. The VP1 capsid protein of adeno-associated virus type 2 is carrying a phospholipase A2 domain required for virus infectivity. The Journal of General Virology. May 2002, 83 (Pt 5): 973–8. PMID 11961250. doi:10.1099/0022-1317-83-5-973
 .
.
- ^ Warrington KH, Gorbatyuk OS, Harrison JK, Opie SR, Zolotukhin S, Muzyczka N. Adeno-associated virus type 2 VP2 capsid protein is nonessential and can tolerate large peptide insertions at its N terminus. Journal of Virology. June 2004, 78 (12): 6595–609. PMC 416546
 . PMID 15163751. doi:10.1128/JVI.78.12.6595-6609.2004.
. PMID 15163751. doi:10.1128/JVI.78.12.6595-6609.2004.
- ^ Mori S, Wang L, Takeuchi T, Kanda T. Two novel adeno-associated viruses from cynomolgus monkey: pseudotyping characterization of capsid protein. Virology. December 2004, 330 (2): 375–83. PMID 15567432. doi:10.1016/j.virol.2004.10.012
 .
.
- ^ Weitzman, Matthew D.; Linden, R. Michael. Adeno-Associated Virus Biology. Snyder, R. O.; Moullier, P (编). Adeno-associated virus methods and protocols. Totowa, NJ: Humana Press. 2011. ISBN 978-1-61779-370-7.
- ^ Bartlett JS, Samulski RJ, McCown TJ. Selective and rapid uptake of adeno-associated virus type 2 in brain. Human Gene Therapy. May 1998, 9 (8): 1181–6. PMID 9625257. doi:10.1089/hum.1998.9.8-1181.
- ^ Fischer AC, Beck SE, Smith CI, Laube BL, Askin FB, Guggino SE, Adams RJ, Flotte TR, Guggino WB. Successful transgene expression with serial doses of aerosolized rAAV2 vectors in rhesus macaques. Molecular Therapy. December 2003, 8 (6): 918–26. PMID 14664794. doi:10.1016/j.ymthe.2003.08.015
 .
.
- ^ Nicklin SA, Buening H, Dishart KL, de Alwis M, Girod A, Hacker U, Thrasher AJ, Ali RR, Hallek M, Baker AH. Efficient and selective AAV2-mediated gene transfer directed to human vascular endothelial cells. Molecular Therapy. September 2001, 4 (3): 174–81. PMID 11545607. doi:10.1006/mthe.2001.0424
 .
.
- ^ Qing K, Mah C, Hansen J, Zhou S, Dwarki V, Srivastava A. Human fibroblast growth factor receptor 1 is a co-receptor for infection by adeno-associated virus 2. Nature Medicine. January 1999, 5 (1): 71–7. PMID 9883842. S2CID 31602526. doi:10.1038/4758.
- ^ Summerford C, Samulski RJ. Membrane-associated heparan sulfate proteo-glycan is a receptor for adeno-associated virus type 2 virions. Journal of Virology. Feb 1998, 72 (2): 1438–45. PMC 124624
 . PMID 9445046. doi:10.1128/JVI.72.2.1438-1445.1998.
. PMID 9445046. doi:10.1128/JVI.72.2.1438-1445.1998.
- ^ Summerford C, Bartlett JS, Samulski RJ. AlphaVbeta5 integrin: a co-receptor for adeno-associated virus type 2 infection. Nature Medicine. January 1999, 5 (1): 78–82. PMID 9883843. S2CID 23326070. doi:10.1038/4768.
- ^ Qiu J, Handa A, Kirby M, Brown KE. The interaction of heparin sulfate and adeno-associated virus 2. Virology. March 2000, 269 (1): 137–47. PMID 10725206. doi:10.1006/viro.2000.0205
 .
.
- ^ ICTV Taxonomy history: Adeno-associated dependoparvovirus A. ICTV. [31 January 2020]. (原始内容存档于2021-07-12).
- ^ ICTV Taxonomy history: Adeno-associated dependoparvovirus B. ICTV. [31 January 2020]. (原始内容存档于2021-07-11).
- ^ Adeno-associated Virus (AAV) Guide. Addgene. [2021-07-10]. (原始内容存档于2022-10-05).
- ^ 43.0 43.1 43.2 Worgall, Stefan; Crystal, Ronald G. Gene therapy: 493–518. 2020. doi:10.1016/B978-0-12-818422-6.00029-0.
- ^ Shams, Shahin; Silva, Eduardo A. Bioengineering strategies for gene delivery: 107–148. 2020. doi:10.1016/B978-0-12-816221-7.00004-5.
- ^ Nony P, Tessier J, Chadeuf G, Ward P, Giraud A, Dugast M, Linden RM, Moullier P, Salvetti A. Novel cis-acting replication element in the adeno-associated virus type 2 genome is involved in amplification of integrated rep-cap sequences. Journal of Virology. October 2001, 75 (20): 9991–4. PMC 114572
 . PMID 11559833. doi:10.1128/JVI.75.20.9991-9994.2001.
. PMID 11559833. doi:10.1128/JVI.75.20.9991-9994.2001.
- ^ Nony P, Chadeuf G, Tessier J, Moullier P, Salvetti A. Evidence for packaging of rep-cap sequences into adeno-associated virus (AAV) type 2 capsids in the absence of inverted terminal repeats: a model for generation of rep-positive AAV particles. Journal of Virology. January 2003, 77 (1): 776–81. PMC 140600
 . PMID 12477885. doi:10.1128/JVI.77.1.776-781.2003.
. PMID 12477885. doi:10.1128/JVI.77.1.776-781.2003.
- ^ Philpott NJ, Giraud-Wali C, Dupuis C, Gomos J, Hamilton H, Berns KI, Falck-Pedersen E. Efficient integration of recombinant adeno-associated virus DNA vectors requires a p5-rep sequence in cis. Journal of Virology. June 2002, 76 (11): 5411–21. PMC 137060
 . PMID 11991970. doi:10.1128/JVI.76.11.5411-5421.2002.
. PMID 11991970. doi:10.1128/JVI.76.11.5411-5421.2002.
- ^ Tullis GE, Shenk T. Efficient replication of adeno-associated virus type 2 vectors: a cis-acting element outside of the terminal repeats and a minimal size. Journal of Virology. December 2000, 74 (24): 11511–21. PMC 112431
 . PMID 11090148. doi:10.1128/JVI.74.24.11511-11521.2000.
. PMID 11090148. doi:10.1128/JVI.74.24.11511-11521.2000.
- ^ Vectors used in Gene Therapy Clinical Trials. Journal of Gene Medicine. Wiley. December 2019 [2012-01-04]. (原始内容存档于2019-10-21).
