戊二酰亚胺:修订间差异
删除的内容 添加的内容
小 ItMarki移動頁面User:ItMarki/戊二酰亚胺至戊二酰亚胺 |
补救3个来源,并将0个来源标记为失效。) #IABot (v2.0.9.2 |
||
| 第24行: | 第24行: | ||
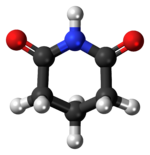
'''戊二酰亚胺'''是一种[[有机化合物]],分子式为(CH<sub>2</sub>)<sub>3</sub>(CO)<sub>2</sub>NH。它是一种白色固体。该化合物在[[戊二酸]]酰胺脱水后形成。<ref>{{Cite journal |last=Paris |first=G. |last2=Berlinguet |first2=L. |last3=Gaudry |first3=R. |last4=English, Jr. |first4=J. |last5=Dayan |first5=J. E. |year=1957 |title=Glutaric Acid and Glutarimide |journal=Organic Syntheses |volume=37 |page=47 |doi=10.15227/orgsyn.037.0047}}</ref> |
'''戊二酰亚胺'''是一种[[有机化合物]],分子式为(CH<sub>2</sub>)<sub>3</sub>(CO)<sub>2</sub>NH。它是一种白色固体。该化合物在[[戊二酸]]酰胺脱水后形成。<ref>{{Cite journal |last=Paris |first=G. |last2=Berlinguet |first2=L. |last3=Gaudry |first3=R. |last4=English, Jr. |first4=J. |last5=Dayan |first5=J. E. |year=1957 |title=Glutaric Acid and Glutarimide |journal=Organic Syntheses |volume=37 |page=47 |doi=10.15227/orgsyn.037.0047}}</ref> |
||
戊二酰亚胺有时称作2,6-哌啶二酮。它是多种[[药物]]的核心,包括[[沙利度胺]],一种用于治疗[[多发性骨髓瘤]]<ref>{{Cite web |title=A to Z List of Cancer Drugs: Thalidomide |url=https://www.cancer.gov/about-cancer/treatment/drugs/thalidomide |publisher=National Cancer Institute |access-date=20 September 2021}}</ref>和[[麻风病]]的药物;<ref name="nyt-fda">{{Cite news|url=https://www.nytimes.com/1998/07/17/us/thalidomide-approved-to-treat-leprosy-with-other-uses-seen.html|title=Thalidomide Approved to Treat Leprosy, With Other Uses Seen|work=New York Times|first=Sheryl Gay|author=Stolberg|accessdate=20 September 2021|date=17 July 1998}}</ref>和[[放线菌酮]],一种有效的蛋白质合成抑制剂。<ref>Hugh D. Sisler, Malcolm R. Siegel; [https://link.springer.com/chapter/10.1007/978-3-642-46051-7_21 Cycloheximide and Other Glutarimide Antibiotics]; Mechanism of Action pp 283-307</ref> |
戊二酰亚胺有时称作2,6-哌啶二酮。它是多种[[药物]]的核心,包括[[沙利度胺]],一种用于治疗[[多发性骨髓瘤]]<ref>{{Cite web |title=A to Z List of Cancer Drugs: Thalidomide |url=https://www.cancer.gov/about-cancer/treatment/drugs/thalidomide |publisher=National Cancer Institute |access-date=20 September 2021 |archive-date=2022-08-15 |archive-url=https://web.archive.org/web/20220815180447/https://www.cancer.gov/about-cancer/treatment/drugs/thalidomide |dead-url=no }}</ref>和[[麻风病]]的药物;<ref name="nyt-fda">{{Cite news|url=https://www.nytimes.com/1998/07/17/us/thalidomide-approved-to-treat-leprosy-with-other-uses-seen.html|title=Thalidomide Approved to Treat Leprosy, With Other Uses Seen|work=New York Times|first=Sheryl Gay|author=Stolberg|accessdate=20 September 2021|date=17 July 1998|archive-date=2020-12-03|archive-url=https://web.archive.org/web/20201203003055/https://www.nytimes.com/1998/07/17/us/thalidomide-approved-to-treat-leprosy-with-other-uses-seen.html|dead-url=no}}</ref>和[[放线菌酮]],一种有效的蛋白质合成抑制剂。<ref>Hugh D. Sisler, Malcolm R. Siegel; [https://link.springer.com/chapter/10.1007/978-3-642-46051-7_21 Cycloheximide and Other Glutarimide Antibiotics] {{Wayback|url=https://link.springer.com/chapter/10.1007/978-3-642-46051-7_21 |date=20220804175044 }}; Mechanism of Action pp 283-307</ref> |
||
== 参考文献 == |
== 参考文献 == |
||
2022年10月21日 (五) 22:41的版本
| 戊二酰亚胺 | |
|---|---|

| |
| |
| IUPAC名 Piperidine-2,6-dione 哌啶-2,6-二酮 | |
| 别名 | 2,6-Diketopiperidine |
| 识别 | |
| CAS号 | 1121-89-7 |
| PubChem | 70726 |
| ChemSpider | 63891 |
| SMILES |
|
| 性质 | |
| 化学式 | C5H7NO2 |
| 摩尔质量 | 113.11 g/mol g·mol⁻¹ |
| 熔点 | 155-157 °C[1] |
| 若非注明,所有数据均出自标准状态(25 ℃,100 kPa)下。 | |
戊二酰亚胺是一种有机化合物,分子式为(CH2)3(CO)2NH。它是一种白色固体。该化合物在戊二酸酰胺脱水后形成。[2]
戊二酰亚胺有时称作2,6-哌啶二酮。它是多种药物的核心,包括沙利度胺,一种用于治疗多发性骨髓瘤[3]和麻风病的药物;[4]和放线菌酮,一种有效的蛋白质合成抑制剂。[5]
参考文献
- ^ Glutarimide - Sigma-Aldrich
- ^ Paris, G.; Berlinguet, L.; Gaudry, R.; English, Jr., J.; Dayan, J. E. Glutaric Acid and Glutarimide. Organic Syntheses. 1957, 37: 47. doi:10.15227/orgsyn.037.0047.
- ^ A to Z List of Cancer Drugs: Thalidomide. National Cancer Institute. [20 September 2021]. (原始内容存档于2022-08-15).
- ^ Stolberg, Sheryl Gay. Thalidomide Approved to Treat Leprosy, With Other Uses Seen. New York Times. 17 July 1998 [20 September 2021]. (原始内容存档于2020-12-03).
- ^ Hugh D. Sisler, Malcolm R. Siegel; Cycloheximide and Other Glutarimide Antibiotics (页面存档备份,存于互联网档案馆); Mechanism of Action pp 283-307
