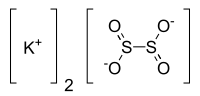
連二亞硫酸鉀是一種無機化合物,化學式為K2S2O4。它的UN編號為1929。它可由三仲丁基硼氫化鉀和二氧化硫在四氫呋喃中反應製得;[1]它也可由連二亞硫酸鋅和氫氧化鉀反應得到。[2]在氟化鉀存在下,它可以將硝酸鈾醯還原為KxUIVFy(如K7U6F31等)。[2]它和硫化鉀、硫和鈦在650 °C反應,可以得到硫代鈦酸鹽K6Ti2OS6。[3]
參考文獻[編輯]
- ^ Steven A. Chmielewski, Dennis W. Bennett. A New Synthon for Main Group Metal Dithionites in a Non-Aqueous Environment. Synthesis and Reactivity in Inorganic and Metal-Organic Chemistry. 1986-01, 16 (1): 95–103 [2022-09-21]. ISSN 0094-5714. doi:10.1080/00945718608055914 (英語).
- ^ 2.0 2.1 Richard A. Hermens, Paul L. Driver, Casey V. Pedro, Xue Rong Zhang, Stephen C. Schlegel, Jeff M. Burning. Synthesis of metal fluorouranate(IV) compounds in aqueous solution. Journal of Fluorine Chemistry. 1996-03, 77 (1): 13–14 [2022-09-21]. doi:10.1016/0022-1139(95)03367-X (英語).
- ^ Klepp, Kurt O.; Fabian, Ferdinand. Preparation and crystal structure of hexapotassium-μ-oxo-hexathiodititanate(IV), K6Ti2OS6. Zeitschrift fuer Naturforschung, B: Chemical Sciences, 2003. 58 (2/3): 163-167. CODEN ZNBSEN
|
|---|
| | 稀有氣體、IA、IIA | |
|---|
| | 硼族、碳族 | |
|---|
| | 氮族 | |
|---|
| | 氧族 | |
|---|
| | 鹵素、擬鹵素 | |
|---|
| | 副族金屬 | |
|---|
| | 有機酸鹽 | |
|---|
|