User:Kevin88lue/昆蟲形態學

昆蟲形態學是一門研究和描述昆蟲身體結構的學科,由於昆蟲和其他節肢動物共享部分演化史,昆蟲形態學的術語會和節肢動物形態學類似。昆蟲的特徵包括:體分三節(頭部、胸部及腹部)、三對足和位於頭殼外的口器,以上三個特徵是區別昆蟲及其他節肢動物重要特徵。六足亞門包含原尾目、雙尾目、彈尾目及昆蟲,前三者與昆蟲的區別特徵就是口器的位置。
昆蟲種類眾多,其身體結構變異多端,體型小至 0.3 mm(缨小蜂),大至 30 cm(長喙天蛾)[1]:7;複眼有或無;翅膀發達或無翅;足特化成奔跑用、跳躍用、游泳用及挖掘用,昆蟲藉著這些特化的構造佔據了深海與南極洲之外所有的生態區位。本文將會描述昆蟲身體的基本結構和幾種常見的特化,本文也將會定義數個用以描述昆蟲形態的術語。
解剖學總述[编辑]
昆蟲和其他節肢動物一樣沒有內骨骼,它們只有由幾丁質組成的外骨骼,用來保護並支撐整個身體。昆蟲的身體可以區分成三個區塊:頭部、胸部和腹部[2],頭部負責感知外界和攝取食物;胸部是足和翅膀(若有翅)的著生點,特化成適合運動的結構;腹部則負責消化、呼吸、分泌,和繁殖[1]:22–48。雖然這三個區塊的功能大致上相同,但基礎結構卻形態各形態各異,翅膀、足、觸角和口器在不同類群中差異甚大[3]。
External[编辑]
外骨骼[编辑]
昆蟲的外骨骼,也就是「表皮(cuticle)」,由兩層構成:上表皮(epicuticle)及原表皮(procuticle),上表皮位於最外側,是一層薄薄的含蠟防水層,不含幾丁質(chitin),上表皮的下方是原表皮,原表皮含有幾丁質,比上表皮厚,可再下分為兩層:靠外層的外表皮(exocuticle)及靠內層的內表皮(endocuticle),外表皮骨化(sclerotized),質地堅硬不易變形,內表皮則由幾丁質纖維及蛋白質縱橫交錯編織,一層一層如三明治般堆疊而成,質地韌,彈性十足[1]:22–24。很多身體較軟的昆蟲外表皮含量較少,尤其是幼蟲期(如毛毛蟲)。幾丁質是N-乙醯葡糖胺的長鏈聚合物,一種葡萄糖的衍生物。未修飾的幾丁質顏色透明,堅韌、易彎曲、富含彈性,在節肢動物上出現的幾丁質多數是經修飾的,它們被鑲入硬化的蛋白質矩陣,形成了所謂的外骨骼,單純的外骨骼質地類似皮革,但在表面加上碳酸鈣之後,質地會變得更加堅硬[4]。毛毛蟲的外骨骼未經修飾,質地較為柔軟,甲蟲則經過修飾,質地堅硬,未修飾及修飾後的差異可以從兩者比較。
外部的表皮及內部的基底膜由一層柱狀的上皮細胞組成,昆蟲大部分的身體構造皆被內表皮所包覆,表皮能夠支持並保護蟲體,然而,表皮沒辦法隨著蟲體長大,因此昆蟲每隔一段時間就必須把外部骨化的表皮蛻下,在快要脫皮的時候,大部分的外表皮會被溶解並吸收。脫皮的過程中,首先舊表皮從真皮層脫落(apolysis),舊表皮和真皮層間會充滿含有分解酶的液體,液體會分解內表皮並將其合成為新的表皮,外表皮不會被分解,當新表皮生長到足夠的程度,上表皮及殘餘的外表皮會一起脫落,此過程稱為「蛻皮(ecdysis)」[5]:16–20。
昆蟲的身體可以大致分成四個區域,背面(dorsal)、腹面(ventral)及兩個側面(laterals),也可稱為背側(tergum)、腹側(sternum)及側邊(pleura)。外骨骼上硬化的板狀結構稱之為骨片(sclerites),分別為背板(tergites)、腹板(sternites)及側板(pleurites)[6]。
頭部[编辑]



大部分昆蟲的頭部由高度骨化的堅硬外骨骼包住,也就是「頭殼(head capsule)」。多數的例外為未完全骨化的幼蟲,特別是完全變態的昆蟲,但就算是身體未骨化或骨化程度弱的幼蟲都生有發達的骨化頭殼,如甲蟲和膜翅目的幼蟲。環裂群(Cyclorrhapha)的幼蟲則幾乎不具頭殼。
大部分的感官皆生於頭殼上,如觸角(antenna)、單眼(ocellus)、複眼(compound eyes)及口器(mouthparts),成蟲的頭殼沒有分節,這點是無庸置疑的,但是經過胚胎學的研究後發現,頭殼分成六節,分別生有成對的副肢,如口器,口器的每個部份都生在特定的頭殼分節上,一個分節上只會有一對副肢,但在現生的昆蟲中,不是每個分節都一定生有副肢。
在所有的昆蟲中,直翅目的頭部變異最為多元,其縫線(suture)和骨片(sclerite)變化多端[6]。下口式(hypognathous)及後口式(opisthognathous)昆蟲的頭頂(vertex)位於複眼之間,在頭的背側,而前口式(prognathous)昆蟲的頭頂則在單眼的位置,這是因為前口式昆蟲的頭部旋轉了 90°,變得和體軸平行。某些昆蟲的頭頂會特化,學者因此給予它們不同的名字[7]:13。
蛻皮線(ecdsial suture)又稱頭縫線(epicranial suture),由冠縫線 (coronal suture) 及前額縫線(frontal suture)組成,蛻皮線變異極大,在不同的昆蟲中形態相異。蛻皮線始於頭頂,垂直地向下延伸並分叉,將前額上方的區域劃分為兩塊。蛻皮線的形狀隨物種而異,可以是Y型、U型,甚至是V型。蛻皮線中分叉的線條稱之為前額縫線或額頰縫線(frontogenal suture),不是每種昆蟲都具有前額縫線,在蛻皮時,前額縫線會裂開,形成能讓昆蟲離開舊皮的開口。
頭頂下方的區域為前額(frons),前額的大小因物種而異,在很多昆蟲中,前額的界線往往是模糊不清的,連某些頭殼發達的物種也不例外。在大部分的物種中,前額的下方的界線為額楯溝或口上溝,側邊的界線為額頰溝,上方的界線則為蛻皮線。如果昆蟲生有中央單眼,十之八九都位於前額上,但在某些昆蟲中,如膜翅目昆蟲,三顆單眼都位於頭頂。前額比較明確的定義為:咽擴張肌著生的骨片,但在大部分情況下,這個定義往往派不上用場[8]。在某些類群中,如蟬形下目(Cicadomorpha),前額寬大且近乎垂直,因而易於辨認,頭部的中央區域及為前額[9]。
頭楯是介於前額和上唇之間的骨片,在原始昆蟲中,頭楯的上緣和前額由額楯溝(frontoclypeal suture)區隔。頭楯側緣的邊界由楯頰溝(clypeogenal suture)界定,下緣則由楯上唇溝(clypeolabral suture)界定。頭楯的形狀因物種而異,如鱗翅目擁有很大的頭楯部和很長的口器。頰部 (gena) 為頭部兩側的骨化區域,位於複眼下方,複眼下方延伸出一條頰縫線 (gular suture)。頰部的形狀因物種而異,因此其界限難以定義,例如:在蜻蛉目中,頰部位於複眼、頭楯部和口器之間。後頰部 (postgena) 位於頰部的正後方,在有翅亞綱中,後頰部位於頰部的後方或下方。後頭弓 (occipital arch) 是頭殼後側一個狹窄的區塊,呈彎曲狀,蓋住後頭孔 (foramen)。下頰部 (subgenal) 通常很狹窄,位於口器之上,下唇板 (hypostoma) 和側唇板 (pleurostoma) 也位於下頰部[7]:13–14。頭頂 (vertex) 向後延伸,越過觸角基部,形狀類似倒T形。頭殼後方生有後頭孔,內部組織透過後頭孔將頭部與身體連結,如神經節、食道、唾腺、肌肉組織等[10]。
On the posterior aspect of the head are the occiput, postgena, occipital foramen, posterior tentorial pit, gula, postgenal bridge, hypostomal suture and bridge, and the mandibles, labium, and maxilla. The occipital suture is well founded in species of Orthoptera, but not so much in other orders. Where found, the occipital suture is the arched, horseshoe-shaped groove on the back of the head, ending at the posterior of each mandible. The postoccipital suture is a landmark on the posterior surface of the head, and is typically near the occipital foremen. In pterygotes, the postocciput forms the extreme posterior, often U-shaped, which forms the rim of the head extending to the postoccipital suture. In pterygotes, such as those of Orthoptera, the occipital foramen and the mouth are not separated. The three types of occipital closures, or points under the occipital foramen that separate the two lower halves of the postgena, are: the hypostomal bridge, the postgenal bridge, and the gula. The hypostomal bridge is usually found in insects with hypognathous orientation. The postgenal bridge is found in the adults of species of higher Diptera and aculeate Hymenoptera, while the gula is found on some Coleoptera, Neuroptera, and Isoptera, which typically display prognathous-oriented mouthparts.[7]:15
複眼和單眼[编辑]
大多數的昆蟲都具有複眼 (compound eyes),複眼由小眼 (ommatidia,單數形為 ommatidium) 組成,小眼的數量因物種而異,例如蜻蜓的複眼由高達三萬個小眼組成。複眼的解析度比脊椎動物的眼睛低,但複眼能精準的感知動作,且通常能接收紫外光和綠光,有些物種還能接受其他光譜的光。通常還能夠感知偏振光[11]。通常還會有兩至三顆外凸的單眼 (ocelli),每顆單眼面向的方向不同。複眼能看見的角度的角度比單眼廣,解析度也較高,但有些幼蟲的單眼功能與複眼不相上下,如 Tenthredinidae 科的幼蟲單眼敏銳度為4度,且對於偏振光的感知度極高[12][13]。
Because the individual lenses are so small, the effects of diffraction impose a limit on the possible resolution that can be obtained (assuming they do not function as phased arrays). This can only be countered by increasing lens size and number. To see with a resolution comparable to our simple eyes, humans would require compound eyes that would each reach the size of their heads. Compound eyes fall into two groups: apposition eyes, which form multiple inverted images, and superposition eyes, which form a single erect image.[14][15] Compound eyes grow at their margins by the addition of new ommatidia.[16]
觸角[编辑]

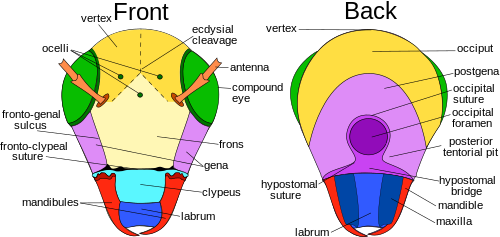
觸角是一對靈活的副肢,位於昆蟲的頭部,用來感知環境。觸角上面佈滿了細緻的剛毛 (setae),昆蟲利用這些剛毛來感知環境[17]:8–11。除了觸覺以外,昆蟲還能透過觸角上微小的感覺結構來接受氣味、感覺氣溫、濕度、壓力,甚至是本體感覺[17]:8–11[18][19]。有些昆蟲還能透過觸角感知聲波,如蜜蜂和某些蠅類[20]。
觸角的節數因昆蟲種類而異,高等蠅類 (higher flies) 僅有3-6節[21],而蟑螂成蟲可超過140節[22]。觸角形狀的種類也族繁不及備載,觸角的第一節(連結在頭部的那節)稱作柄節 (scape),第二節稱作梗節 (pedicel),其他的節稱作鞭節 (flagellum)[17]:8–11。
以下為幾種常見的觸角形狀:
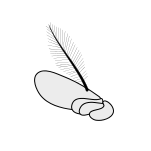 芒狀 (Aristate) 芒狀 (Aristate)
|
 Capitate Capitate
|
 棒狀 (Clavate) 棒狀 (Clavate)
|
 絲狀 (Filiform) 絲狀 (Filiform)
|
 Flabellate Flabellate
|
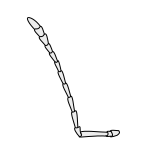 膝狀 (Geniculate) 膝狀 (Geniculate)
|
 Setaceous Setaceous
|
 扇形 (Lamellate) 扇形 (Lamellate)
|
 念珠狀 (Moniliform) 念珠狀 (Moniliform)
|
 梳狀 (Pectinate) 梳狀 (Pectinate)
|
 羽狀 (Plumose) 羽狀 (Plumose)
|
 鋸齒狀 (Serrate) 鋸齒狀 (Serrate)
|
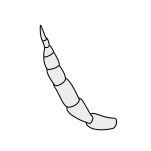 Stylate Stylate
|
Mouthparts[编辑]
昆蟲的口器由數個部分組成,分別為上唇 (labrum)、下唇 (labium)、大顎 (mandibles)、小顎 (maxillae,單數型為 maxilla)[7]:16[23]。上唇為一構造簡單的骨片,可上下移動,上唇與頭楯相連,上唇移動時可帶動頭楯 (clypeus)。大顎為一對高度骨化的構造,運動方向與體軸垂直,用以啃咬、咀嚼或切斷食物。小顎成對,運動方向亦與體軸垂直,小顎生有分節的附肢,為小顎鬚 (maxillary palps)。下唇為縱向運動的構造,生有一對分節的附肢,為下唇鬚 (labial palps)[24]。

口器依照與身體的相對位置能分成三種類型,分別為:前口式 (prognathous)、下口式 (hypognathous) 和後口式 (opisthognathous)。前口式的昆蟲口器與體軸平行,如黑豔蟲 (Passalidae),下口式的昆蟲口器與體軸垂直,如螞蟻 (Formicidae),後口式的昆蟲口器與體軸平行,但口器向身體後方延伸,如蜚蠊目 (Blattodea)[25]。口器的形態與功能因物種而異,共可粗分為兩種,具顎式 (mandibulate) 與吸吮式 (haustellate)。吸吮式口器可用於吸取液體,可依照口針 (stylet) 有無進一步區分,有口針的可再分成刺吸式 (piercing-sucking)、吸吮式 (sponging) 和虹吸式 (siphoning) 等。口針為一針狀構造,用以刺穿植物或動物組織。口針和攝食道 (feeding tube) 由大顎、小顎及下咽特化而來[24]。
- 咀嚼式(或具顎式,Mandibulate ):最常見的類型,用以鉗咬敵人和咬碎食物。
- 刺吸式 (Piercing-sucking):具有口針,能刺穿固體並吸取液體食物。
- 吸吮式 (Sponging):用以舔取液體食物,無口針(大多數雙翅目口器為此類)
- 虹吸式 (Siphoning):用以吸取液體食物,無口針(大多數鱗翅目口器為此類)
擁有咀嚼式口器的昆蟲包括:蜻蛉目、脈翅目成蟲、鞘翅目、膜翅目、蜚蠊目、直翅目和鱗翅目幼蟲。
咀嚼式[编辑]
上唇連接於頭楯的下方,為蓋住口腔的寬葉[1]:22–24。上唇的內側為膜質,有些物種中央會生有上咽 (epipharynx),上咽生有感覺器 (sensilla),感覺器可分為物理感覺器 (mechanosensilla) 與化學感覺器 (chemosensilla)[26][27]。上唇依靠兩束肌肉支撐,這兩束肌肉始於頭部內,並漸漸朝中央靠攏,最後伸入上唇前緣側後方兩個骨片,舌側片 (tormae),在某些昆蟲中,負責連結頭楯帶動上唇的表皮含有彈性蛋白,使頭楯移動更為靈活[28]。以前認為上唇與第一頭節相關,然而,近期的胚胎發育學、基因表現與神經的研究顯示,上唇是由第三頭節的第三腦 (tritocerebrum) 控制,顯示上唇是由第三頭節的附肢演化而來[1]:22–24。
Chewing insects have two mandibles, one on each side of the head. The mandibles are positioned between the labrum and maxillae. The mandibles cut and crush food, and may be used for defense; generally, they have an apical cutting edge, and the more basal molar area grinds the food. They can be extremely hard (around 3 on Mohs, or an indentation hardness of about 30 kg/mm2); thus, many termites and beetles have no physical difficulty in boring through foils made from such common metals as copper, lead, tin, and zinc.[1]:22–24 The cutting edges are typically strengthened by the addition of zinc, manganese, or rarely, iron, in amounts up to about 4% of the dry weight.[26] They are typically the largest mouthparts of chewing insects, being used to masticate (cut, tear, crush, chew) food items. They open outwards (to the sides of the head) and come together medially. In carnivorous, chewing insects, the mandibles can be modified to be more knife-like, whereas in herbivorous chewing insects, they are more typically broad and flat on their opposing faces (e.g., caterpillars). In male stag beetles, the mandibles are modified to such an extent as to not serve any feeding function, but are instead are used to defend mating sites from other males. In ants, the mandibles also serve a defensive function (particularly in soldier castes). In bull ants, the mandibles are elongated and toothed, used as hunting (and defensive) appendages.
Situated beneath the mandibles, paired maxillae manipulate food during mastication. Maxillae can have hairs and "teeth" along their inner margins. At the outer margin, the galea is a cupped or scoop-like structure, which sits over the outer edge of the labium. They also have palps, which are used to sense the characteristics of potential foods. The maxillae occupy a lateral position, one on each side of the head behind the mandibles. The proximal part of the maxilla consists of a basal cardo, which has a single articulation with the head, and a flat plate, the stipes, hinged to the cardo. Both cardo and stipes are loosely joined to the head by membrane so they are capable of movement. Distally on the stipes are two lobes, an inner lacinea and an outer galea, one or both of which may be absent. More laterally on the stipes is a jointed, leglike palp made up of a number of segments; in Orthoptera there are five. Anterior and posterior rotator muscles are inserted on the cardo, and ventral adductor muscles arising on the tentorium are inserted on both cardo and stipes. Arising in the stipes are flexor muscles of lacinea and galea and another lacineal flexor arises in the cranium, but neither the lacinea nor the galea has an extensor muscle. The palp has levator and depressor muscles arising in the stipes, and each segment of the palp has a single muscle causing flexion of the next segment.[28]
In mandibulate mouthparts, the labium is a quadrupedal structure, although it is formed from two fused secondary maxillae. It can be described as the floor of the mouth. With the maxillae, it assists with manipulation of food during mastication or chewing or, in the unusual case of the dragonfly nymph, extends out to snatch prey back to the head, where the mandibles can eat it. The labium is similar in structure to the maxilla, but with the appendages of the two sides fused by the midline, so they come to form a median plate. The basal part of the labium, equivalent to the maxillary cardines and possibly including a part of the sternum of the labial segment, is called the postmentum. This may be subdivided into a proximal submentum and a distal mentum. Distal to the postmentum, and equivalent to the fused maxillary stipites, is the prementum. The prementum closes the preoral cavity from behind. Terminally, it bears four lobes, two inner glossae, and two outer paraglossae, which are collectively known as the ligula. One or both pairs of lobes may be absent or they may be fused to form a single median process. A palp arises from each side of the prementum, often being three-segmented.[28]
The hypopharynx is a median lobe immediately behind the mouth, projecting forwards from the back of the preoral cavity; it is a lobe of uncertain origin, but perhaps associated with the mandibular segment;[28] in apterygotes, earwigs, and nymphal mayflies, the hypopharynx bears a pair of lateral lobes, the superlinguae (singular: superlingua). It divides the cavity into a dorsal food pouch, or cibarium, and a ventral salivarium into which the salivary duct opens.[1]:22–24 It is commonly found fused to the libium.[26] Most of the hypopharynx is membranous, but the adoral face is sclerotized distally, and proximally contains a pair of suspensory sclerites extending upwards to end in the lateral wall of the stomodeum. Muscles arising on the frons are inserted into these sclerites, which distally are hinged to a pair of lingual sclerites. These, in turn, have inserted into them antagonistic pairs of muscles arising on the tentorium and labium. The various muscles serve to swing the hypopharynx forwards and back, and in the cockroach, two more muscles run across the hypopharynx and dilate the salivary orifice and expand the salivarium.[28]
刺吸式[编辑]
口器有很多種功能,有些昆蟲的口器結合穿刺和汲取,刺穿生物組織再吸取液體。雌蚊取食血液,為血食性 (hemophagous),此習性使它們為病媒。蚊子口氣由喙 (proboscis)、大顎及小顎組成。小顎特化為口針,口針被包裹在下唇內,蚊子取食時,小顎能刺穿皮膚和固定口器。包裹口器的下唇向後滑動,其餘的口器露出並進入皮膚,接著蚊子透過下咽注射包含抗凝血劑的唾液,最後,蚊子用上唇吸取血液。瘧蚊屬的小顎鬚很長,幾乎等同於上唇[29]。
Siphoning[编辑]
The proboscis is formed from maxillary galeae and is adaption found in some insects for sucking.[30] The muscles of the cibarium or pharynx are strongly developed and form the pump. In Hemiptera and many Diptera, which feed on fluids within plants or animals, some components of the mouthparts are modified for piercing, and the elongated structures are called stylets. The combined tubular structures are referred to as the proboscis, although specialized terminology is used in some groups.
In species of Lepidoptera, it consists of two tubes held together by hooks and separable for cleaning. Each tube is inwardly concave, thus forming a central tube through which moisture is sucked. Suction is effected through the contraction and expansion of a sac in the head.[31] The proboscis is coiled under the head when the insect is at rest, and is extended only when feeding.[30] The maxillary palpi are reduced or even vestigial.[32] They are conspicuous and five-segmented in some of the more basal families, and are often folded.[7] The shape and dimensions of the proboscis have evolved to give different species wider and therefore more advantageous diets.[30] There is an allometric scaling relationship between body mass of Lepidoptera and length of proboscis[33] from which an interesting adaptive departure is the unusually long-tongued hawk moth Xanthopan morganii praedicta. Charles Darwin predicted the existence and proboscis length of this moth before its discovery based on his knowledge of the long-spurred Madagascan star orchid Angraecum sesquipedale.[34]
Sponging[编辑]
The mouthparts of insects that feed on fluids are modified in various ways to form a tube through which liquid can be drawn into the mouth and usually another through which saliva passes. The muscles of the cibarium or pharynx are strongly developed to form a pump.[28] In nonbiting flies, the mandibles are absent and other structures are reduced; the labial palps have become modified to form the labellum, and the maxillary palps are present, although sometimes short. In Brachycera, the labellum is especially prominent and used for sponging liquid or semiliquid food.[35] The labella are a complex structure consisting of many grooves, called pseudotracheae, which sop up liquids. Salivary secretions from the labella assist in dissolving and collecting food particles so they can be more easily taken up by the pseudotracheae; this is thought to occur by capillary action. The liquid food is then drawn up from the pseudotracheae through the food channel into the esophagus.[36]
The mouthparts of bees are of a chewing and lapping-sucking type. Lapping is a mode of feeding in which liquid or semiliquid food adhering to a protrusible organ, or "tongue", is transferred from substrate to mouth. In the honey bee (Hymenoptera: Apidae: Apis mellifera), the elongated and fused labial glossae form a hairy tongue, which is surrounded by the maxillary galeae and the labial palps to form a tubular proboscis containing a food canal. In feeding, the tongue is dipped into the nectar or honey, which adheres to the hairs, and then is retracted so the adhering liquid is carried into the space between the galeae and labial palps. This back-and-forth glossal movement occurs repeatedly. Movement of liquid to the mouth apparently results from the action of the cibarial pump, facilitated by each retraction of the tongue pushing liquid up the food canal.[1]:22–24
Thorax[编辑]
The insect thorax has three segments: the prothorax, mesothorax, and metathorax. The anterior segment, closest to the head, is the prothorax; its major features are the first pair of legs and the pronotum. The middle segment is the mesothorax; its major features are the second pair of legs and the anterior wings, if any. The third, the posterior, thoracic segment, abutting the abdomen, is the metathorax, which bears the third pair of legs and the posterior wings. Each segment is dilineated by an intersegmental suture. Each segment has four basic regions. The dorsal surface is called the tergum (or notum, to distinguish it from the abdominal terga).[1]:22–24 The two lateral regions are called the pleura (singular: pleuron), and the ventral aspect is called the sternum. In turn, the notum of the prothorax is called the pronotum, the notum for the mesothorax is called the mesonotum and the notum for the metathorax is called the metanotum. Continuing with this logic, there is also the mesopleura and metapleura, as well as the mesosternum and metasternum.[7]
The tergal plates of the thorax are simple structures in apterygotes and in many immature insects, but are variously modified in winged adults. The pterothoracic nota each have two main divisions: the anterior, wing-bearing alinotum and the posterior, phragma-bearing postnotum. Phragmata (singular: phragma) are plate-like apodemes that extend inwards below the antecostal sutures, marking the primary intersegmental folds between segments; phragmata provide attachment for the longitudinal flight muscles. Each alinotum (sometimes confusingly referred to as a "notum") may be traversed by sutures that mark the position of internal strengthening ridges, and commonly divides the plate into three areas: the anterior prescutum, the scutum, and the smaller posterior scutellum. The lateral pleural sclerites are believed to be derived from the subcoxal segment of the ancestral insect leg. These sclerites may be separate, as in silverfish, or fused into an almost continuous sclerotic area, as in most winged insects.[1]:22–24
前胸[编辑]
相較於其他胸版,前胸較小,構造也較為簡單,但某些類群的前胸發達(如甲蟲、螳螂、椿象和某些直翅目),甚至在蜚蠊目中,前胸極為發達且蓋住部分的頭部和中胸[7][1]:22–24。
Pterothorax[编辑]
Because the mesothorax and metathorax hold the wings, they have a combined name called the pterothorax (pteron = wing). The forewing, which goes by different names in different orders (e.g., the tegmina in Orthoptera and elytra in Coleoptera), arises between the mesonotum and the mesopleuron, and the hindwing articulates between the metanotum and metapleuron. The legs arise from the mesopleuron and metapleura. The mesothorax and metathorax each have a pleural suture (mesopleural and metapleural sutures) that runs from the wing base to the coxa of the leg. The sclerite anterior to the pleural suture is called the episternum (serially, the mesepisternum and metepisternum). The sclerite posterior to the suture is called the epimiron (serially, the mesepimiron and metepimiron). Spiracles, the external organs of the respiratory system, are found on the pterothorax, usually one between the pro- and mesopleoron, as well as one between the meso- and metapleuron.[7]
The ventral view or sternum follows the same convention, with the prosternum under the prothorax, the mesosternum under the mesothorax and the metasternum under the metathorax. The notum, pleura, and sternum of each segment have a variety of different sclerites and sutures, varying greatly from order to order, and they will not be discussed in detail in this section.[7]
Wings[编辑]
Most phylogenetically advanced insects have two pairs of wings located on the second and third thoracic segments.[1]:22–24 Insects are the only invertebrates to have developed flight capability, and this has played an important part in their success. Insect flight is not very well understood, relying heavily on turbulent aerodynamic effects. The primitive insect groups use muscles that act directly on the wing structure. The more advanced groups making up the Neoptera have foldable wings, and their muscles act on the thorax wall and power the wings indirectly.[1]:22–24 These muscles are able to contract multiple times for each single nerve impulse, allowing the wings to beat faster than would ordinarily be possible.
Insect flight can be extremely fast, maneuverable, and versatile, possibly due to the changing shape, extraordinary control, and variable motion of the insect wing. Insect orders use different flight mechanisms; for example, the flight of a butterfly can be explained using steady-state, nontransitory aerodynamics, and thin airfoil theory.
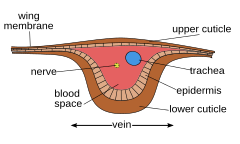
Internal[编辑]
Each of the wings consists of a thin membrane supported by a system of veins. The membrane is formed by two layers of integument closely apposed, while the veins are formed where the two layers remain separate and the cuticle may be thicker and more heavily sclerotized. Within each of the major veins is a nerve and a trachea, and, since the cavities of the veins are connected with the hemocoel, hemolymph can flow into the wings.[28] Also, the wing lumen, being an extension of the hemocoel, contains the tracheae, nerves, and hemolymph. As the wing develops, the dorsal and ventral integumental layers become closely apposed over most of their area, forming the wing membrane. The remaining areas form channels, the future veins, in which the nerves and tracheae may occur. The cuticle surrounding the veins becomes thickened and more heavily sclerotized to provide strength and rigidity to the wing. Hairs of two types may occur on the wings: microtrichia, which are small and irregularly scattered, and macrotrichia, which are larger, socketed, and may be restricted to veins. The scales of Lepidoptera and Trichoptera are highly modified macrotrichia.[26]
Veins[编辑]

In some very small insects, the venation may be greatly reduced. In chalcid wasps, for instance, only the subcosta and part of the radius are present. Conversely, an increase in venation may occur by the branching of existing veins to produce accessory veins or by the development of additional, intercalary veins between the original ones, as in the wings of Orthoptera (grasshoppers and crickets). Large numbers of cross-veins are present in some insects, and they may form a reticulum as in the wings of Odonata (dragonflies and damselflies) and at the base of the forewings of Tettigonioidea and Acridoidea (katydids and grasshoppers, respectively).[28]
The archedictyon is the name given to a hypothetical scheme of wing venation proposed for the very first winged insect. It is based on a combination of speculation and fossil data. Since all winged insects are believed to have evolved from a common ancestor, the archediction represents the "template" that has been modified (and streamlined) by natural selection for 200 million years. According to current dogma, the archedictyon contained six to eight longitudinal veins. These veins (and their branches) are named according to a system devised by John Comstock and George Needham—the Comstock-Needham system:[37]
- Costa (C) - the leading edge of the wing
- Subcosta (Sc) - second longitudinal vein (behind the costa), typically unbranched
- Radius (R) - third longitudinal vein, one to five branches reach the wing margin
- Media (M) - fourth longitudinal vein, one to four branches reach the wing margin
- Cubitus (Cu) - fifth longitudinal vein, one to three branches reach the wing margin
- Anal veins (A1, A2, A3) - unbranched veins behind the cubitus
The costa (C) is the leading marginal vein on most insects, although a small vein, the precosta, is sometimes found above the costa. In almost all extant insects,[1]:41–42 the precosta is fused with the costa; the costa rarely ever branches because it is at the leading edge, which is associated at its base with the humeral plate. The trachea of the costal vein is perhaps a branch of the subcostal trachea. Located after the costa is the third vein, the subcosta, which branches into two separate veins: the anterior and posterior. The base of the subcosta is associated with the distal end of the neck of the first axillary. The fourth vein is the radius, which is branched into five separate veins. The radius is generally the strongest vein of the wing. Toward the middle of the wing, it forks into a first undivided branch (R1) and a second branch, called the radial sector (Ra), which subdivides dichotomously into four distal branches (R2, R3, R4, R5). Basally, the radius is flexibly united with the anterior end of the second axillary (2Ax).[38]
The fifth vein of the wing is the media. In the archetype pattern (A), the media forks into two main branches, a media anterior (MA), which divides into two distal branches (MA1, MA2), and a median sector, or media posterior (MP), which has four terminal branches (M1, M2, M3, M4). In most modern insects, the media anterior has been lost, and the usual "media" is the four-branched media posterior with the common basal stem. In the Ephemerida, according to present interpretations of the wing venation, both branches of the media are retained, while in Odonata, the persisting media is the primitive anterior branch. The stem of the media is often united with the radius, but when it occurs as a distinct vein, its base is associated with the distal median plate (m') or is continuously sclerotized with the latter. The cubitus, the sixth vein of the wing, is primarily two-branched. The primary forking takes place near the base of the wing, forming the two principal branches (Cu1, Cu2). The anterior branch may break up into a number of secondary branches, but commonly it forks into two distal branches. The second branch of the cubitus (Cu2) in Hymenoptera, Trichoptera, and Lepidoptera, was mistaken by Comstock and Needham for the first anal. Proximally, the main stem of the cubitus is associated with the distal median plate (m') of the wing base.[38]
The postcubitus (Pcu) is the first anal of the Comstock and Needham system. The postcubitus, however, has the status of an independent wing vein and should be recognized as such. In nymphal wings, its trachea arises between the cubital trachea and the group of vannal tracheae. In the mature wings of more generalized insects, the postcubitus is always associated proximally with the cubitus, and is never intimately connected with the flexor sclerite (3Ax) of the wing base. In Neuroptera, Mecoptera, and Trichoptera, the postcubitus may be more closely associated with the vannal veins, but its base is always free from the latter. The postcubitus is usually unbranched; primitively, it is two-branched. The vannal veins (lV to nV) are the anal veins immediately associated with the third axillary, and which are directly affected by the movement of this sclerite that brings about the flexion of the wings. In number, the vannal veins vary from one to 12, according to the expansion of the vannal area of the wing. The vannal tracheae usually arise from a common tracheal stem in nymphal insects, and the veins are regarded as branches of a single anal vein. Distally, the vannal veins are either simple or branched. The jugal vein (J) of the jugal lobe of the wing is often occupied by a network of irregular veins, or it may be entirely membranous; sometimes it contains one or two distinct, small veins, the first jugal vein, or vena arcuata, and the second jugal vein, or vena cardinalis (2J).[38]
- C-Sc cross-veins - run between the costa and subcosta
- R cross-veins - run between adjacent branches of the radius
- R-M cross-veins - run between the radius and media
- M-Cu cross-veins - run between the media and cubitus
All the veins of the wing are subject to secondary forking and to union by cross-veins. In some orders of insects, the cross-veins are so numerous, the whole venational pattern becomes a close network of branching veins and cross-veins. Ordinarily, however, a definite number of cross-veins having specific locations occurs. The more constant cross-veins are the humeral cross-vein (h) between the costa and subcosta, the radial cross-vein (r) between R and the first fork of Rs, the sectorial cross-vein (s) between the two forks of R8, the median cross-vein (m-m) between M2 and M3, and the mediocubital cross-vein (m-cu) between the media and the cubitus.[38]
The veins of insect wings are characterized by a convex-concave placement, such as those seen in mayflies (i.e., concave is "down" and convex is "up"), which alternate regularly and by their branching; whenever a vein forks there is always an interpolated vein of the opposite position between the two branches. The concave vein will fork into two concave veins (with the interpolated vein being convex) and the regular alteration of the veins is preserved.[39] The veins of the wing appear to fall into an undulating pattern according to whether they have a tendency to fold up or down when the wing is relaxed. The basal shafts of the veins are convex, but each vein forks distally into an anterior convex branch and a posterior concave branch. Thus, the costa and subcosta are regarded as convex and concave branches of a primary first vein, Rs is the concave branch of the radius, posterior media the concave branch of the media, Cu1 and Cu2 are respectively convex and concave, while the primitive postcubitus and the first vannal have each an anterior convex branch and a posterior concave branch. The convex or concave nature of the veins has been used as evidence in determining the identities of the persisting distal branches of the veins of modern insects, but it has not been demonstrated to be consistent for all wings.[28][38]
Fields[编辑]

Wing areas are delimited and subdivided by fold lines, along which the wings can fold, and flexion lines, which flex during flight. Between the flexion and the fold lines, the fundamental distinction is often blurred, as fold lines may permit some flexibility or vice versa. Two constants, found in nearly all insect wings, are the claval (a flexion line) and jugal folds (or fold line), forming variable and unsatisfactory boundaries. Wing foldings can very complicated, with transverse folding occurring in the hindwings of Dermaptera and Coleoptera, and in some insects, the anal area can be folded like a fan.[1]:41–42 The four different fields found on insect wings are:
- Remigium
- Anal area (vannus)
- Jugal area
- Axillary area
- Alula
Most veins and cross-veins occur in the anterior area of the remigium, which is responsible for most of the flight, powered by the thoracic muscles. The posterior portion of the remigium is sometimes called the clavus[需要消歧义]; the two other posterior fields are the anal and jugal areas.[1]:41–42 When the vannal fold has the usual position anterior to the group of anal veins, the remigium contains the costal, subcostal, radial, medial, cubital, and postcubital veins. In the flexed wing, the remigium turns posteriorly on the flexible basal connection of the radius with the second axillary, and the base of the mediocubital field is folded medially on the axillary region along the plica basalis (bf) between the median plates (m, m') of the wing base.[38]
The vannus is bordered by the vannal fold, which typically occurs between the postcubitus and the first vannal vein. In Orthoptera, it usually has this position. In the forewing of Blattidae, however, the only fold in this part of the wing lies immediately before the postcubitus. In Plecoptera, the vannal fold is posterior to the postcubitus, but proximally it crosses the base of the first vannal vein. In the cicada, the vannal fold lies immediately behind the first vannal vein (lV). These small variations in the actual position of the vannal fold, however, do not affect the unity of action of the vannal veins, controlled by the flexor sclerite (3Ax), in the flexion of the wing. In the hindwings of most Orthoptera, a secondary vena dividens forms a rib in the vannal fold. The vannus is usually triangular in shape, and its veins typically spread out from the third axillary like the ribs of a fan. Some of the vannal veins may be branched, and secondary veins may alternate with the primary veins. The vannal region is usually best developed in the hindwing, in which it may be enlarged to form a sustaining surface, as in Plecoptera and Orthoptera. The great fan-like expansions of the hindwings of Acrididae are clearly the vannal regions, since their veins are all supported on the third axillary sclerites on the wing bases, though Martynov (1925) ascribes most of the fan areas in Acrididae to the jugal regions of the wings. The true jugum of the acridid wing is represented only by the small membrane (Ju) mesad of the last vannal vein. The jugum is more highly developed in some other Orthoptera, as in the Mantidae. In most of the higher insects with narrow wings, the vannus becomes reduced, and the vannal fold is lost, but even in such cases, the flexed wing may bend along a line between the postcubitus and the first vannal vein.[38]
The jugal region, or neala, is a region of the wing that is usually a small membranous area proximal to the base of the vannus strengthened by a few small, irregular vein-like thickenings; but when well developed, it is a distinct section of the wing and may contain one or two jugal veins. When the jugal area of the forewing is developed as a free lobe, it projects beneath the humeral angle of the hindwing and thus serves to yoke the two wings together. In the Jugatae group of Lepidoptera, it bears a long finger-like lobe. The jugal region was termed the neala ("new wing") because it is evidently a secondary and recently developed part of the wing.[38]
The auxiliary region containing the axillary sclerites has, in general, the form of a scalene triangle. The base of the triangle (a-b) is the hinge of the wing with the body; the apex (c) is the distal end of the third axillary sclerite; the longer side is anterior to the apex. The point d on the anterior side of the triangle marks the articulation of the radial vein with the second axillary sclerite. The line between d and c is the plica basalis (bf), or fold of the wing at the base of the mediocubital field.[38]
At the posterior angle of the wing base in some Diptera there is a pair of membranous lobes (squamae, or calypteres) known as the alula. The alula is well developed in the house fly. The outer squama (c) arises from the wing base behind the third axillary sclerite (3Ax) and evidently represents the jugal lobe of other insects (A, D); the larger inner squama (d) arises from the posterior scutellar margin of the tergum of the wing-bearing segment and forms a protective, hood-like canopy over the halter. In the flexed wing, the outer squama of the alula is turned upside down above the inner squama, the latter not being affected by the movement of the wing. In many Diptera, a deep incision of the anal area of the wing membrane behind the single vannal vein sets off a proximal alar lobe distal to the outer squama of the alula.[38]
Joints[编辑]

The various movements of the wings, especially in insects that flex their wings horizontally over their backs when at rest, demand a more complicated articular structure at the wing base than a mere hinge of the wing with the body. Each wing is attached to the body by a membranous basal area, but the articular membrane contains a number of small articular sclerites, collectively known as the pteralia. The pteralia include an anterior humeral plate at the base of the costal vein, a group of axillaries (Ax) associated with the subcostal, radial, and vannal veins, and two less definite median plates (m, m') at the base of the mediocubital area. The axillaries are specifically developed only in the wing-flexing insects, where they constitute the flexor mechanism of the wing operated by the flexor muscle arising on the pleuron. Characteristic of the wing base is also a small lobe on the anterior margin of the articular area proximal to the humeral plate, which, in the forewing of some insects, is developed into a large, flat, scale-like flap, the tegula, overlapping the base of the wing. Posteriorly, the articular membrane often forms an ample lobe between the wing and the body, and its margin is generally thickened and corrugated, giving the appearance of a ligament, the so-called axillary cord, continuous mesally with the posterior marginal scutellar fold of the tergal plate bearing the wing.[38]
The articular sclerites, or pteralia, of the wing base of the wing-flexing insects and their relations to the body and the wing veins, shown diagrammatically, are as follows:
- Humeral plates
- First Axillary
- Second Axillary
- Third Axillary
- Fourth Axillary
- Median plates (m, m')
The humeral plate is usually a small sclerite on the anterior margin of the wing base, movable and articulated with the base of the costal vein. Odonata have their humeral plates greatly enlargened,[38] with two muscles arising from the episternum inserted into the humeral plates and two from the edge of the epimeron inserted into the axillary plate.[28]
The first axillary sclerite (lAx) is the anterior hinge plate of the wing base. Its anterior part is supported on the anterior notal wing process of the tergum (ANP); its posterior part articulates with the tergal margin. The anterior end of the sclerite is generally produced as a slender arm, the apex of which (e) is always associated with the base of the subcostal vein (Sc), though it is not united with the latter. The body of the sclerite articulates laterally with the second axillary. The second axillary sclerite (2Ax) is more variable in form than the first axillary, but its mechanical relations are no less definite. It is obliquely hinged to the outer margin of the body of the first axillary, and the radial vein (R) is always flexibly attached to its anterior end (d). The second axillary presents both a dorsal and a ventral sclerotization in the wing base; its ventral surface rests upon the fulcral wing process of the pleuron. The second axillary, therefore, is the pivotal sclerite of the wing base, and it specifically manipulates the radial vein.[38]
The third axillary sclerite (3Ax) lies in the posterior part of the articular region of the wing. Its form is highly variable and often irregular, but the third axillary is the sclerite on which is inserted the flexor muscle of the wing (D). Mesally, it articulates anteriorly (f) with the posterior end of the second axillary, and posteriorly (b) with the posterior wing process of the tergum (PNP), or with a small fourth axillary when the latter is present. Distally, the third axillary is prolonged in a process always associated with the bases of the group of veins in the anal region of the wing, here termed the vannal veins (V). The third axillary, therefore, is usually the posterior hinge plate of the wing base and is the active sclerite of the flexor mechanism, which directly manipulates the vannal veins. The contraction of the flexor muscle (D) revolves the third axillary on its mesal articulations (b, f), and thereby lifts its distal arm; this movement produces the flexion of the wing. The fourth axillary sclerite is not a constant element of the wing base. When present, it is usually a small plate intervening between the third axillary and the posterior notal wing process, and is probably a detached piece of the latter.[38]
The median plates (m, m') are also sclerites that are not so definitely differentiated as specific plates as are the three principal axillaries, but they are important elements of the flexor apparatus. They lie in the median area of the wing base distal to the second and third axillaries, and are separated from each other by an oblique line (bf), which forms a prominent convex fold during flexion of the wing. The proximal plate (m) is usually attached to the distal arm of the third axillary and perhaps should be regarded as a part of the latter. The distal plate (m') is less constantly present as a distinct sclerite, and may be represented by a general sclerotization of the base of the mediocubital field of the wing. When the veins of this region are distinct at their bases, they are associated with the outer median plate.[38]
Coupling, folding, and other features[编辑]
In many insect species, the forewing and hindwing are coupled together, which improves the aerodynamic efficiency of flight. The most common coupling mechanism (e.g., Hymenoptera and Trichoptera) is a row of small hooks on the forward margin of the hindwing, or "hamuli", which lock onto the forewing, keeping them held together (hamulate coupling). In some other insect species (e.g., Mecoptera, Lepidoptera, and some Trichoptera) the jugal lobe of the forewing covers a portion of the hindwing (jugal coupling), or the margins of the forewing and hindwing overlap broadly (amplexiform coupling), or the hindwing bristles, or frenulum, hook under the retaining structure or retinalucum on the forewing.[1]:43
When at rest, the wings are held over the back in most insects, which may involve longitudinal folding of the wing membrane and sometimes also transverse folding. Folding may sometimes occur along the flexion lines. Though fold lines may be transverse, as in the hindwings of beetles and earwigs, they are normally radial to the base of the wing, allowing adjacent sections of a wing to be folded over or under each other. The commonest fold line is the jugal fold, situated just behind the third anal vein,[26] although, most Neoptera have a jugal fold just behind vein 3A on the forewings. It is sometimes also present on the hindwings. Where the anal area of the hindwing is large, as in Orthoptera and Blattodea, the whole of this part may be folded under the anterior part of the wing along a vannal fold a little posterior to the claval furrow. In addition, in Orthoptera and Blattodea, the anal area is folded like a fan along the veins, the anal veins being convex, at the crests of the folds, and the accessory veins concave. Whereas the claval furrow and jugal fold are probably homologous in different species, the vannal fold varies in position in different taxa. Folding is produced by a muscle arising on the pleuron and inserted into the third axillary sclerite in such a waythat, when it contracts, the sclerite pivots about its points of articulation with the posterior notal process and the second axillary sclerite.[28]
As a result, the distal arm of the third axillary sclerite rotates upwards and inwards, so that finally its position is completely reversed. The anal veins are articulated with this sclerite in such a way that when it moves they are carried with it and become flexed over the back of the insect. Activity of the same muscle in flight affects the power output of the wing and so it is also important in flight control. In orthopteroid insects, the elasticity of the cuticle causes the vannal area of the wing to fold along the veins. Consequently, energy is expended in unfolding this region when the wings are moved to the flight position. In general, wing extension probably results from the contraction of muscles attached to the basalar sclerite or, in some insects, to the subalar sclerite.[28]
Legs[编辑]
The typical and usual segments of the insect leg are divided into the coxa, one trochanter, the femur, the tibia, the tarsus, and the pretarsus. The coxa in its more symmetrical form, has the shape of a short cylinder or truncate cone, though commonly it is ovate and may be almost spherical. The proximal end of the coxa is girdled by a submarginal basicostal suture that forms internally a ridge, or basicosta, and sets off a marginal flange, the coxomarginale, or basicoxite. The basicosta strengthens the base of the coxa and is commonly enlarged on the outer wall to give insertion to muscles; on the mesal half of the coxa, however, it is usually weak and often confluent with the coxal margin. The trochanteral muscles that take their origin in the coxa are always attached distal to the basicosta. The coxa is attached to the body by an articular membrane, the coxal corium, which surrounds its base. These two articulations are perhaps the primary dorsal and ventral articular points of the subcoxo-coxal hinge. In addition, the insect coxa has often an anterior articulation with the anterior, ventral end of the trochantin, but the trochantinal articulation does not coexist with a sternal articulation. The pleural articular surface of the coxa is borne on a mesal inflection of the coxal wall. If the coxa is movable on the pleural articulation alone, the coxal articular surface is usually inflected to a sufficient depth to give a leverage to the abductor muscles inserted on the outer rim of the coxal base. Distally the coxa bears an anterior and a posterior articulation with the trochanter. The outer wall of the coxa is often marked by a suture extending from the base to the anterior trochanteral articulation. In some insects the coxal suture falls in line with the pleural suture, and in such cases the coxa appears to be divided into two parts corresponding to the episternum and epimeron of the pleuron. The coxal suture is absent in many insects.[38]:163–164
The inflection of the coxal wall bearing the pleural articular surface divides the lateral wall of the basicoxite into a prearticular part and a postarticular part, and the two areas often appear as two marginal lobes on the base of the coxa. The posterior lobe is usually the larger and is termed the meron. The meron may be greatly enlarged by an extension distally in the posterior wall of the coxa; in the Neuroptera, Mecoptera, Trichoptera, and Lepidoptera, the meron is so large that the coxa appears to be divided into an anterior piece, the so-called "coxa genuina," and the meron, but the meron never includes the region of the posterior trochanteral articulation, and the groove delimiting it is always a part of the basicostal suture. A coxa with an enlarged meron has an appearance similar to one divided by a coxal suture falling in line with the pleural suture, but the two conditions are fundamentally quite different and should not be confused. The meron reaches the extreme of its departure from the usual condition in the Diptera. In some of the more generalized flies, as in the Tipulidae, the meron of the middle leg appears as a large lobe of the coxa projecting upward and posteriorly from the coxal base; in higher members of the order it becomes completely separated from the coxa and forms a plate of the lateral wall of the mesothorax.[38]:164
The trochanter is the basal segment of the telopodite; it is always a small segment in the insect leg, freely movable by a horizontal hinge on the coxa, but more or less fixed to the base of the femur. When movable on the femur the trochantero femoral hinge is usually vertical or oblique in a vertical plane, giving a slight movement of production and reduction at the joint, though only a reductor muscle is present. In the Odonata, both nymphs and adults, there are two trochanteral segments, but they are not movable on each other; the second contains the reductor muscle of the femur. The usual single trochanteral segment of insects, therefore, probably represents the two trochanters of other arthropods fused into one apparent segment, since it is not likely that the primary coxotrochanteral hinge has been lost from the leg. In some of the Hymenoptera a basal subdivision of the femur simulates a second trochanter, but the insertion of the reductor muscle on its base attests that it belongs to the femoral segment, since as shown in the odonate leg, the reductor has its origin in the true second trochanter.[38]:165
The femur is the third segment of the insect leg, is usually the longest and strongest part of the limb, but it varies in size from the huge hind femur of leaping Orthoptera to a very small segment such as is present in many larval forms. The volume of the femur is generally correlated with the size of the tibial muscles contained within it, but it is sometimes enlarged and modified in shape for other purposes than that of accommodating the tibial muscles. The tibia is characteristically a slender segment in adult insects, only a little shorter than the femur or the combined femur and trochanter. Its proximal end forms a more or less distinct head bent toward the femur, a device allowing the tibia to be flexed close against the under surface of the femur.[38]:165
The terms profemur, mesofemur and metafemur refer to the femora of the front, middle and hind legs of an insect, respectively.[40] Similarly protibia, mesotibia and metatibia refer to the tibiae of the front, middle and hind legs.[41]
The tarsus of insects corresponds to the penultimate segment of a generalized arthropod limb, which is the segment called the propodite in Crustacea. adult insects it is commonly subdivided into from two to five subsegments, or tarsomeres, but in the Protura, some Collembola, and most holometabolous insect larvae it preserves the primitive form of a simple segment. The subsegments of the adult insect tarsus are usually freely movable on one another by inflected connecting membranes, but the tarsus never has intrinsic muscles. The tarsus of adult pterygote insects having fewer than five subsegments is probably specialized by the loss of one or more subsegments or by a fusion of adjoining subsegments. In the tarsi of Acrididae the long basal piece is evidently composed of three united tarsomeres, leaving the fourth and the fifth. The basal tarsomere is sometimes conspicuously enlarged and is distinguished as the basitarsus. On the under surfaces of the tarsal subsegments in certain Orthoptera there are small pads, the tarsal pulvilli, or euplantulae. The tarsus is occasionally fused with the tibia in larval insects, forming a tibiotarsal segment; in some cases it appears to be eliminated or reduced to a rudiment between the tibia and the pretarsus.[38]:165–166
For the most part the femur and tibia are the longest leg segments but variations in the lengths and robustness of each segment relate to their functions. For example, gressorial and cursorial, or walking and running type insects respectively, usually have well-developed femora and tibiae on all legs, whereas jumping (saltatorial) insects such as grasshoppers have disproportionately developed metafemora and metatibiae. In aquatic beetles (Coleoptera) and bugs (Hemiptera), the tibiae and/or tarsi of one or more pairs of legs usually are modified for swimming (natatorial) with fringes of long, slender hairs. Many ground-dwelling insects, such as mole crickets (Orthoptera: Gryllotalpidae), nymphal cicadas (Hemiptera: Cicadidae), and scarab beetles (Scarabaeidae), have the tibiae of the forelegs (protibiae) enlarged and modified for digging (fossorial), whereas the forelegs of some predatory insects, such as mantispid lacewings (Neuroptera) and mantids (Mantodea), are specialized for seizing prey, or raptorial. The tibia and basal tarsomere of each hindleg of honey bees are modified for the collection and carriage of pollen.[28]:45
Abdomen[编辑]
The ground plan of the abdomen of an adult insect typically consists of 11–12 segments and is less strongly sclerotized than the head or thorax. Each segment of the abdomen is represented by a sclerotized tergum, sternum, and perhaps a pleurite. Terga are separated from each other and from the adjacent sterna or pleura by a membrane. Spiracles are located in the pleural area. Variation of this ground plan includes the fusion of terga or terga and sterna to form continuous dorsal or ventral shields or a conical tube. Some insects bear a sclerite in the pleural area called a laterotergite. Ventral sclerites are sometimes called laterosternites. During the embryonic stage of many insects and the postembryonic stage of primitive insects, 11 abdominal segments are present. In modern insects there is a tendency toward reduction in the number of the abdominal segments, but the primitive number of 11 is maintained during embryogenesis.Variation in abdominal segment number is considerable. If the Apterygota are considered to be indicative of the ground plan for pterygotes, confusion reigns: adult Protura have 12 segments, Collembola have 6. The orthopteran family Acrididae has 11 segments, and a fossil specimen of Zoraptera has a 10-segmented abdomen.[7]
Generally, the first seven abdominal segments of adults (the pregenital segments) are similar in structure and lack appendages. However, apterygotes (bristletails and silverfish) and many immature aquatic insects have abdominal appendages. Apterygotes possess a pair of styles; rudimentary appendages that are serially homologous with the distal part of the thoracic legs. And, mesally, one or two pairs of protrusible (or exsertile) vesicles on at least some abdominal segments. These vesicles are derived from the coxal and trochanteral endites (inner annulated lobes) of the ancestral abdominal appendages. Aquatic larvae and nymphs may have gills laterally on some to most abdominal segments.[1]:49 Of the rest of the abdominal segments consist of the reproductive and anal parts.
The anal-genital part of the abdomen, known as the terminalia, consists generally of segments 8 or 9 to the abdominal apex. Segments 8 and 9 bear the genitalia; segment 10 is visible as a complete segment in many "lower" insects but always lacks appendages; and the small segment 11 is represented by a dorsal epiproct and pair of ventral paraprocts derived from the sternum. A pair of appendages, the cerci, articulates laterally on segment 11; typically these are annulated and filamentous but have been modified (e.g. the forceps of earwigs) or reduced in different insect orders. An annulated caudal filament, the median appendix dorsalis, arises from the tip of the epiproct in apterygotes, most mayflies (Ephemeroptera), and a few fossil insects. A similar structure in nymphal stoneflies (Plecoptera) is of uncertain homology. These terminal abdominal segments have excretory and sensory functions in all insects, but in adults there is an additional reproductive function.[1]:49
External genitalia[编辑]

The organs concerned specifically with mating and the deposition of eggs are known collectively as the external genitalia, although they may be largely internal. The components of the external genitalia of insects are very diverse in form and often have considerable taxonomic value, particularly among species that appear structurally similar in other respects. The male external genitalia have been used widely to aid in distinguishing species, whereas the female external genitalia may be simpler and less varied.
The terminalia of adult female insects include internal structures for receiving the male copulatory organ and his spermatozoa and external structures used for oviposition (egg-laying; section 5.8). Most female insects have an egg-laying tube, or ovipositor; it is absent in termites, parasitic lice, many Plecoptera, and most Ephemeroptera. Ovipositors take two forms:
- true, or appendicular, formed from appendages of abdominal segments 8 and 9;
- substitutional, composed of extensible posterior abdominal segments.
Other appendages[编辑]
Internal[编辑]
Nervous system[编辑]
The nervous system of an insect can be divided into a brain and a ventral nerve cord. The head capsule is made up of six fused segments, each with a pair of ganglia, or a cluster of nerve cells outside of the brain. The first three pairs of ganglia are fused into the brain, while the three following pairs are fused into a structure of three pairs of ganglia under the insect's esophagus, called the subesophageal ganglion.[1]:57
The thoracic segments have one ganglion on each side, which are connected into a pair, one pair per segment. This arrangement is also seen in the abdomen but only in the first eight segments. Many species of insects have reduced numbers of ganglia due to fusion or reduction.[42] Some cockroaches have just six ganglia in the abdomen, whereas the wasp Vespa crabro has only two in the thorax and three in the abdomen. Some insects, like the house fly Musca domestica, have all the body ganglia fused into a single large thoracic ganglion.
At least a few insects have nociceptors, cells that detect and transmit sensations of pain.[43] This was discovered in 2003 by studying the variation in reactions of larvae of the common fruitfly Drosophila to the touch of a heated probe and an unheated one. The larvae reacted to the touch of the heated probe with a stereotypical rolling behavior that was not exhibited when the larvae were touched by the unheated probe.[44] Although nociception has been demonstrated in insects, there is not a consensus that insects feel pain consciously.[45]
Digestive system[编辑]
An insect uses its digestive system to extract nutrients and other substances from the food it consumes.[46] Most of this food is ingested in the form of macromolecules and other complex substances like proteins, polysaccharides, fats, and nucleic acids. These macromolecules must be broken down by catabolic reactions into smaller molecules like amino acids and simple sugars before being used by cells of the body for energy, growth, or reproduction. This break-down process is known as digestion. The main structure of an insect's digestive system is a long enclosed tube called the alimentary canal, which runs lengthwise through the body. The alimentary canal directs food in one direction: from the mouth to the anus. It has three sections, each of which performs a different process of digestion. In addition to the alimentary canal, insects also have paired salivary glands and salivary reservoirs. These structures usually reside in the thorax, adjacent to the foregut.[1]:70–77 The gut is where almost all of insects' digestion takes place. It can be divided into the foregut, midgut and hindgut.
Foregut[编辑]

The first section of the alimentary canal is the foregut (element 27 in numbered diagram), or stomodaeum. The foregut is lined with a cuticular lining made of chitin and proteins as protection from tough food. The foregut includes the buccal cavity (mouth), pharynx, esophagus, and Crop and proventriculus (any part may be highly modified), which both store food and signify when to continue passing onward to the midgut.[1]:70 Here, digestion starts as partially chewed food is broken down by saliva from the salivary glands. As the salivary glands produce fluid and carbohydrate-digesting enzymes (mostly amylases), strong muscles in the pharynx pump fluid into the buccal cavity, lubricating the food like the salivarium does, and helping blood feeders, and xylem and phloem feeders.
From there, the pharynx passes food to the esophagus, which could be just a simple tube passing it on to the crop and proventriculus, and then on ward to the midgut, as in most insects. Alternately, the foregut may expand into a very enlarged crop and proventriculus, or the crop could just be a diverticulum, or fluid filled structure, as in some Diptera species.[47]:30–31

The salivary glands (element 30 in numbered diagram) in an insect's mouth produce saliva. The salivary ducts lead from the glands to the reservoirs and then forward through the head to an opening called the salivarium, located behind the hypopharynx. By moving its mouthparts (element 32 in numbered diagram) the insect can mix its food with saliva. The mixture of saliva and food then travels through the salivary tubes into the mouth, where it begins to break down.[46][48] Some insects, like flies, have extra-oral digestion. Insects using extra-oral digestion expel digestive enzymes onto their food to break it down. This strategy allows insects to extract a significant proportion of the available nutrients from the food source.[47]:31
中腸[编辑]
食物通過嗉囊後會進入中腸 (Midgut 或 mesenteron)(頁首圖中的13號),大部分的食物在中腸被消化。中腸的腸壁上有微小的突起,稱作微絨毛 (microvilli) ,微絨毛能增加表面積以吸收養分,微絨毛通常生於中腸的前端。微絨毛的位置與功能因物種而異,例如能分泌消化酶的特化微絨毛生於中腸的末端,而負責吸收養分的則生於中腸前端[47]:32。
Hindgut[编辑]
In the hindgut (element 16 in numbered diagram), or proctodaeum, undigested food particles are joined by uric acid to form fecal pellets. The rectum absorbs 90% of the water in these fecal pellets, and the dry pellet is then eliminated through the anus (element 17), completing the process of digestion. The uric acid is formed using hemolymph waste products diffused from the Malpighian tubules (element 20). It is then emptied directly into the alimentary canal, at the junction between the midgut and hindgut. The number of Malpighian tubules possessed by a given insect varies between species, ranging from only two tubules in some insects to over 100 tubules in others.[1]:71–72, 78–80
Respiratory systems[编辑]
Insect respiration is accomplished without lungs. Instead, the insect respiratory system uses a system of internal tubes and sacs through which gases either diffuse or are actively pumped, delivering oxygen directly to tissues that need it via their trachea (element 8 in numbered diagram). Since oxygen is delivered directly, the circulatory system is not used to carry oxygen, and is therefore greatly reduced. The insect circulatory system has no veins or arteries, and instead consists of little more than a single, perforated dorsal tube that pulses peristaltically. Toward the thorax, the dorsal tube (element 14) divides into chambers and acts like the insect's heart. The opposite end of the dorsal tube is like the aorta of the insect circulating the hemolymph, arthropods' fluid analog of blood, inside the body cavity.[1]:61–65[49] Air is taken in through openings on the sides of the abdomen called spiracles.
There are many different patterns of gas exchange demonstrated by different groups of insects. Gas exchange patterns in insects can range from continuous and diffusive ventilation, to discontinuous gas exchange.[1]:65–68 During continuous gas exchange, oxygen is taken in and carbon dioxide is released in a continuous cycle. In discontinuous gas exchange, however, the insect takes in oxygen while it is active and small amounts of carbon dioxide are released when the insect is at rest.[50] Diffusive ventilation is simply a form of continuous gas exchange that occurs by diffusion rather than physically taking in the oxygen. Some species of insect that are submerged also have adaptations to aid in respiration. As larvae, many insects have gills that can extract oxygen dissolved in water, while others need to rise to the water surface to replenish air supplies, which may be held or trapped in special structures.[51][52]
Circulatory system[编辑]
Insect blood or haemolymph's main function is that of transport and it bathes the insect's body organs. Making up usually less than 25% of an insect's body weight, it transports hormones, nutrients and wastes and has a role in, osmoregulation, temperature control, immunity, storage (water, carbohydrates and fats) and skeletal function. It also plays an essential part in the moulting process.[53][54] An additional role of the haemolymph in some orders, can be that of predatory defence. It can contain unpalatable and malodourous chemicals that will act as a deterrent to predators.[1] Haemolymph contains molecules, ions and cells;[1] regulating chemical exchanges between tissues, haemolymph is encased in the insect body cavity or haemocoel.[1][55] It is transported around the body by combined heart (posterior) and aorta (anterior) pulsations, which are located dorsally just under the surface of the body.[1][53][54] It differs from vertebrate blood in that it doesn't contain any red blood cells and therefore is without high oxygen carrying capacity, and is more similar to lymph found in vertebrates.[1][55]
Body fluids enter through one-way valved ostia, which are openings situated along the length of the combined aorta and heart organ. Pumping of the haemolymph occurs by waves of peristaltic contraction, originating at the body's posterior end, pumping forwards into the dorsal vessel, out via the aorta and then into the head where it flows out into the haemocoel.[1][55] The haemolymph is circulated to the appendages unidirectionally with the aid of muscular pumps or accessory pulsatile organs usually found at the base of the antennae or wings and sometimes in the legs,[1] with pumping rates accelerating with periods of increased activity.[54] Movement of haemolymph is particularly important for thermoregulation in orders such as Odonata, Lepidoptera, Hymenoptera and Diptera.[1]
內分泌系統[编辑]
內分泌系統包括:
1. 神經分泌細胞 (Neurosecretory cells)
2. 心側腺 (Corpora cardiaca)
3. 前胸腺 (Prothoracic glands)
4. 咽側線 (Corpora allata)[56][57]
Reproductive system[编辑]
Female[编辑]
Female insects are able make eggs, receive and store sperm, manipulate sperm from different males, and lay eggs. Their reproductive systems are made up of a pair of ovaries, accessory glands, one or more spermathecae, and ducts connecting these parts. The ovaries make eggs and accessory glands produce the substances to help package and lay the eggs. Spermathecae store sperm for varying periods of time and, along with portions of the oviducts, can control sperm use. The ducts and spermathecae are lined with a cuticle.[7]:880
The ovaries are made up of a number of egg tubes, called ovarioles, which vary in size and number by species. The number of eggs that the insect is able to make vary by the number of ovarioles with the rate that eggs can be developed being also influenced by ovariole design. In meroistic ovaries, the eggs-to-be divide repeatedly and most of the daughter cells become helper cells for a single oocyte in the cluster. In panoistic ovaries, each egg-to-be produced by stem germ cells develops into an oocyte; there are no helper cells from the germ line. Production of eggs by panoistic ovaries tends to be slower than that by meroistic ovaries.[7]:880
Accessory glands or glandular parts of the oviducts produce a variety of substances for sperm maintenance, transport, and fertilization, as well as for protection of eggs. They can produce glue and protective substances for coating eggs or tough coverings for a batch of eggs called oothecae. Spermathecae are tubes or sacs in which sperm can be stored between the time of mating and the time an egg is fertilized. Paternity testing of insects has revealed that some, and probably many, female insects use the spermatheca and various ducts to control or bias sperm used in favor of some males over others.[7]:880
Male[编辑]
The main component of the male reproductive system is the testis, suspended in the body cavity by tracheae and the fat body. The more primitive apterygote insects have a single testis, and in some lepidopterans the two maturing testes are secondarily fused into one structure during the later stages of larval development, although the ducts leading from them remain separate. However, most male insects have a pair of testes, inside of which are sperm tubes or follicles that are enclosed within a membranous sac. The follicles connect to the vas deferens by the vas efferens, and the two tubular vasa deferentia connect to a median ejaculatory duct that leads to the outside. A portion of the vas deferens is often enlarged to form the seminal vesicle, which stores the sperm before they are discharged into the female. The seminal vesicles have glandular linings that secrete nutrients for nourishment and maintenance of the sperm. The ejaculatory duct is derived from an invagination of the epidermal cells during development and, as a result, has a cuticular lining. The terminal portion of the ejaculatory duct may be sclerotized to form the intromittent organ, the aedeagus. The remainder of the male reproductive system is derived from embryonic mesoderm, except for the germ cells, or spermatogonia, which descend from the primordial pole cells very early during embryogenesis.[7]:885 The aedeagus can be quite pronounced or de minimis. The base of the aedeagus may be the partially sclerotized phallotheca, also called the phallosoma or theca. In some species the phallotheca contains a space, called the endosoma (internal holding pouch), into which the tip end of the aedeagus may be withdrawn (retracted). The vas deferens is sometimes drawn into (folded into) the phallotheca together with a seminal vesicle.[58][59]
不同類群的內部形態[编辑]
Blattodea[编辑]
Cockroaches are most common in tropical and subtropical climates. Some species are in close association with human dwellings and widely found around garbage or in the kitchen. Cockroaches are generally omnivorous with the exception of the wood-eating species such as Cryptocercus; these roaches are incapable of digesting cellulose themselves, but have symbiotic relationships with various protozoans and bacteria that digest the cellulose, allowing them to extract the nutrients. The similarity of these symbionts in the genus Cryptocercus to those in termites are such that it has been suggested that they are more closely related to termites than to other cockroaches,[60] and current research strongly supports this hypothesis of relationships.[61] All species studied so far carry the obligate mutualistic endosymbiont bacterium Blattabacterium, with the exception of Nocticola australiensis, an Australian cave dwelling species without eyes, pigment or wings, and which recent genetic studies indicates are very primitive cockroaches.[62][63]
Cockroaches, like all insects, breathe through a system of tubes called tracheae. The tracheae of insects are attached to the spiracles, excluding the head. Thus cockroaches, like all insects, are not dependent on the mouth and windpipe to breathe. The valves open when the CO2 level in the insect rises to a high level; then the CO2 diffuses out of the tracheae to the outside and fresh O2 diffuses in. Unlike in vertebrates that depend on blood for transporting O2 and CO2, the tracheal system brings the air directly to cells, the tracheal tubes branching continually like a tree until their finest divisions, tracheoles, are associated with each cell, allowing gaseous oxygen to dissolve in the cytoplasm lying across the fine cuticle lining of the tracheole. CO2 diffuses out of the cell into the tracheole. While cockroaches do not have lungs and thus do not actively breathe in the vertebrate lung manner, in some very large species the body musculature may contract rhythmically to forcibly move air out and in the spiracles; this may be considered a form of breathing.[64]
Coleoptera[编辑]
The digestive system of beetles is primarily based on plants, which they for the most part feed upon, with mostly the anterior midgut performing digestion. However, in predatory species (e.g., Carabidae) most digestion occurs in the crop by means of midgut enzymes. In Elateridae species, the predatory larvae defecate enzymes on their prey, with digestion being extraorally.[7] The alimentary canal basically comprises a short narrow pharynx, a widened expansion, the crop and a poorly developed gizzard. After there is a midgut, that varies in dimensions between species, with a large amount of cecum, with a hingut, with varying lengths. There are typically four to six Malpighian tubules.[65]
The nervous system in beetles contains all the types found in insects, varying between different species. With three thoracic and seven or eight abdominal ganglia can be distinguished to that in which all the thoracic and abdominal ganglia are fused to form a composite structure. Oxygen is obtained via a tracheal system. Air enters a series of tubes along the body through openings called spiracles, and is then taken into increasingly finer fibers.[7] Pumping movements of the body force the air through the system. Some species of diving beetles (Dytiscidae) carry a bubble of air with them whenever they dive beneath the water surface. This bubble may be held under the elytra or it may be trapped against the body using specialized hairs. The bubble usually covers one or more spiracles so the insect can breathe air from the bubble while submerged. An air bubble provides an insect with only a short-term supply of oxygen, but thanks to its unique physical properties, oxygen will diffuse into the bubble and displacing the nitrogen, called passive diffusion, however the volume of the bubble eventually diminishes and the beetle will have to return to the surface.[66]
Like other insect species, beetles have hemolymph instead of blood. The open circulatory system of the beetle is driven by a tube-like heart attached to the top inside of the thorax.
Different glands specialize for different pheromones produced for finding mates. Pheromones from species of Rutelinea are produced from epithelial cells lining the inner surface of the apical abdominal segments or amino acid based pheromones of Melolonthinae from eversible glands on the abdominal apex. Other species produce different types of pheromones. Dermestids produce esters, and species of Elateridae produce fatty-acid-derived aldehydes and acetates.[7] For means of finding a mate also, fireflies (Lampyridae) utilized modified fat body cells with transparent surfaces backed with reflective uric acid crystals to biosynthetically produce light, or bioluminescence. The light produce is highly efficient, as it is produced by oxidation of luciferin by the enzymes luciferase in the presence of ATP (adenosine triphospate) and oxygen, producing oxyluciferin, carbon dioxide, and light.[7]
A notable number of species have developed special glands that produce chemicals for deterring predators (see Defense and predation). The Ground beetle's (of Carabidae) defensive glands, located at the posterior, produce a variety of hydrocarbons, aldehydes, phenols, quinones, esters, and acids released from an opening at the end of the abdomen. While African carabid beetles (e.g., Anthia some of which used to comprise the genus Thermophilum) employ the same chemicals as ants: formic acid.[31] While Bombardier beetles have well developed, like other carabid beetles, pygidial glands that empty from the lateral edges of the intersegment membranes between the seventh and eighth abdominal segments. The gland is made of two containing chambers. The first holds hydroquinones and hydrogen peroxide, with the second holding just hydrogen peroxide plus catalases. These chemicals mix and result in an explosive ejection, forming temperatures of around 100 C, with the breakdown of hydroquinone to H2 + O2 + quinone, with the O2 propelling the excretion.[7]
Tympanal organs are hearing organs. Such an organ is generally a membrane (tympanum) stretched across a frame backed by an air sac and associated sensory neurons. In the order Coleoptera, tympanal organs have been described in at least two families.[30] Several species of the genus Cicindela in the family Cicindelidae have ears on the dorsal surface of the first abdominal segment beneath the wing; two tribes in the family Dynastinae (Scarabaeidae) have ears just beneath the pronotal shield or neck membrane. The ears of both families are to ultrasonic frequencies, with strong evidence that they function to detect the presence of bats via their ultrasonic echolocation. Even though beetles constitute a large order and live in a variety of niches, examples of hearing is surprisingly lacking in species, though it is likely that most are just undiscovered.[7]
Dermaptera[编辑]
The neuroendocrine system is typical of insects. There is a brain, a subesophageal ganglion, three thoracic ganglia, and six abdominal ganglia. Strong neuron connections connect the neurohemal corpora cardiaca to the brain and frontal ganglion, where the closely related median corpus allatum produces juvenile hormone III in close proximity to the neurohemal dorsal aorta. The digestive system of earwigs is like all other insects, consisting of a fore-, mid-, and hindgut, but earwigs lack gastric caecae which are specialized for digestion in many species of insect. Long, slender (extratory) malpighian tubules can be found between the junction of the mid- and hind gut.[7]
The reproductive system of females consist of paired ovaries, lateral oviducts, spermatheca, and a genital chamber. The lateral ducts are where the eggs leave the body, while the spermatheca is where sperm is stored. Unlike other insects, the gonopore, or genital opening is behind the seventh abdominal segment. The ovaries are primitive in that they are polytrophic (the nurse cells and oocytes alternate along the length of the ovariole). In some species these long ovarioles branch off the lateral duct, while in others, short ovarioles appear around the duct.[7]
Diptera[编辑]
The genitalia of female flies are rotated to a varying degree from the position found in other insects. In some flies this is a temporary rotation during mating, but in others it is a permanent torsion of the organs that occurs during the pupal stage. This torsion may lead to the anus being located below the genitals, or, in the case of 360° torsion, to the sperm duct being wrapped around the gut, despite the external organs being in their usual position. When flies mate, the male initially flies on top of the female, facing in the same direction, but then turns round to face in the opposite direction. This forces the male to lie on its back in order for its genitalia to remain engaged with those of the female, or the torsion of the male genitals allows the male to mate while remaining upright. This leads to flies having more reproduction abilities than most insects and at a much quicker rate. Flies come in great populations due to their ability to mate effectively and in a short period of time especially during the mating season.[67]
The female lays her eggs as close to the food source as possible, and development is very rapid, allowing the larva to consume as much food as possible in a short period of time before transforming into the adult. The eggs hatch immediately after being laid, or the flies are ovoviviparous, with the larva hatching inside the mother.[67] Larval flies, or maggots, have no true legs, and little demarcation between the thorax and abdomen; in the more derived species, the head is not clearly distinguishable from the rest of the body. Maggots are limbless, or else have small prolegs. The eyes and antennae are reduced or absent, and the abdomen also lacks appendages such as cerci. This lack of features is an adaptation to a food-rich environment, such as within rotting organic matter, or as an endoparasite.[67] The pupae take various forms, and in some cases develop inside a silk cocoon. After emerging from the pupa, the adult fly rarely lives more than a few days, and serves mainly to reproduce and to disperse in search of new food sources.
Lepidoptera[编辑]
In reproductive system of butterflies and moths, the male genitalia are complex and unclear. In females there are three types of genitalia based on the relating taxa: monotrysian, exoporian, and dytresian. In the monotrysian type there is an opening on the fused segments of the sterna 9 and 10, which act as insemination and oviposition. In the exoporian type (in Hepaloidae and Mnesarchaeoidea) there are two separate places for insemination and oviposition, both occurring on the same sterna as the monotrysian type, 9/10. In most species the genitalia are flanked by two soft lobes, although they may be specialized and sclerotized in some species for ovipositing in area such as crevices and inside plant tissue.[65] Hormones and the glands that produce them run the development of butterflies and moths as they go through their life cycle, called the endocrine system. The first insect hormone PTTH (Prothoracicotropic hormone) operates the species life cycle and diapause (see the relates section).[68] This hormone is produced by corpora allata and corpora cardiaca, where it is also stored. Some glands are specialized to perform certain task such as producing silk or producing saliva in the palpi.[1]:65, 75 While the corpora cardiaca produce PTTH, the corpora allata also produces jeuvanile hormones, and the prothorocic glands produce moulting hormones.
In the digestive system, the anterior region of the foregut has been modified to form a pharyngial sucking pump as they need it for the food they eat, which are for the most part liquids. An esophagus follows and leads to the posterior of the pharynx and in some species forms a form of crop. The midgut is short and straight, with the hindgut being longer and coiled.[65] Ancestors of lepidopteran species, stemming from Hymenoptera, had midgut ceca, although this is lost in current butterflies and moths. Instead, all the digestive enzymes other than initial digestion, are immobilized at the surface of the midgut cells. In larvae, long-necked and stalked goblet cells are found in the anterior and posterior midgut regions, respectively. In insects, the goblet cells excrete positive potassium ions, which are absorbed from leaves ingested by the larvae. Most butterflies and moths display the usual digestive cycle, however species that have a different diet require adaptations to meet these new demands.[7]:279
In the circulatory system, hemolymph, or insect blood, is used to circulate heat in a form of thermoregulation, where muscles contraction produces heat, which is transferred to the rest of the body when conditions are unfavorable.[69] In lepidopteran species, hemolymph is circulated through the veins in the wings by some form of pulsating organ, either by the heart or by the intake of air into the trachea.[1]:69 Air is taken in through spiracles along the sides of the abdomen and thorax supplying the trachea with oxygen as it goes through the lepidopteran's respiratory system. There are three different tracheae supplying oxygen diffusing oxygen throughout the species body: The dorsal, ventral, and visceral. The dorsal tracheae supply oxygen to the dorsal musculature and vessels, while the ventral tracheae supply the ventral musculature and nerve cord, and the visceral tracheae supply the guts, fat bodies, and gonads.[1]:71, 72
延伸閱讀[编辑]
| 維基物種上的相關信息:Kevin88lue/昆蟲形態學 |
| 维基共享资源中相关的多媒体资源:Kevin88lue/昆蟲形態學 |
| 维基语录上的Insects语录 |
- Morphology (biology)
- Insect physiology
- External morphology of Lepidoptera
- External morphology of Odonata
- Insect ecology
- Insect flight
- Invertebrate
- Insect
- Entomology
- Prehistoric insect
References[编辑]
- ^ 1.00 1.01 1.02 1.03 1.04 1.05 1.06 1.07 1.08 1.09 1.10 1.11 1.12 1.13 1.14 1.15 1.16 1.17 1.18 1.19 1.20 1.21 1.22 1.23 1.24 1.25 1.26 1.27 1.28 1.29 1.30 1.31 1.32 1.33 1.34 1.35 1.36 Gullan, P.J.; P.S. Cranston. The Insects: An Outline of Entomology 3. Oxford: Blackwell Publishing. 2005. ISBN 1-4051-1113-5.
- ^ O. Orkin Insect zoo. The University of Nebraska Department of Entomology. [2009-05-03]. (原始内容存档于2009-06-02).
- ^ Resh, Vincent H.; Cardé, Ring T. Encyclopedia of Insects 2nd. San DIego, CA: Academic Press. 2009: 12.
- ^ Campbell, N. A. (1996) Biology (4th edition) Benjamin Cummings, New Work. p. 69 ISBN 0-8053-1957-3
- ^ Gene Kritsky. (2002). A Survey of Entomology. iUniverse. ISBN 978-0-595-22143-1.
- ^ 6.0 6.1 external morphology of Insects (PDF). [2011-03-20]. (原始内容 (PDF)存档于2011-07-19).
- ^ 7.00 7.01 7.02 7.03 7.04 7.05 7.06 7.07 7.08 7.09 7.10 7.11 7.12 7.13 7.14 7.15 7.16 7.17 7.18 7.19 7.20 7.21 7.22 Resh, Vincent H.; Ring T. Carde. Encyclopedia of Insects 2. U. S. A.: Academic Press. July 1, 2009. ISBN 0-12-374144-0.
- ^ Richards, O. W.; Davies, R.G. Imms' General Textbook of Entomology: Volume 1: Structure, Physiology and Development Volume 2: Classification and Biology. Berlin: Springer. 1977. ISBN 0-412-61390-5.
- ^ Smith, John Bernhard, Explanation of terms used in entomology Publisher: Brooklyn entomological society 1906 (May be downloaded from: https://archive.org/details/explanationofter00smit)
- ^ Fox, Richard. External Anatomy. Lander University. 6 Oct 2006 [2011-03-20]. (原始内容存档于2011-03-14).
- ^ Archived copy (PDF). [2011-03-20]. (原始内容 (PDF)存档于2008-10-01).
- ^ Meyer-Rochow, V.B. Structure and function of the larval eye of the sawfly larva Perga. Journal of Insect Physiology. 1974, 20 (8): 1565–1591. PMID 4854430. doi:10.1016/0022-1910(74)90087-0.
- ^ Völkel, R.; Eisner, M.; Weible, K.J. Miniaturized imaging systems. Microelectronic Engineering. 2003, 67–68: 461–472. ISSN 0167-9317. doi:10.1016/S0167-9317(03)00102-3.
- ^ Gaten, Edward. Optics and phylogeny: is there an insight? The evolution of superposition eyes in the Decapoda (Crustacea). Contributions to Zoology. 1998, 67 (4): 223–236. (原始内容存档于January 12, 2013).
- ^ Ritchie, Alexander. Ainiktozoon loganense Scourfield, a protochordate? from the Silurian of Scotland. Alcheringa. 1985, 9 (2): 137. doi:10.1080/03115518508618961.
- ^ Mayer, G. Structure and development of onychophoran eyes: What is the ancestral visual organ in arthropods?. Arthropod Structure and Development. 2006, 35 (4): 231–245. PMID 18089073. doi:10.1016/j.asd.2006.06.003.
- ^ 17.0 17.1 17.2 17.3 Chapman, R.F. The Insects: Structure and Function 4th. Cambridge, UK: Cambridge University Press. 1998. ISBN 0521570484.
- ^ Krause, A.F.; Winkler, A.; Dürr, V. Central drive and proprioceptive control of antennal movements in the walking stick insect. Journal of Physiology, Paris. 2013, 107: 116–129.
- ^ Okada, J; Toh, Y. Peripheral representation of antennal orientation by the scapal hair plate of the cockroach Periplaneta americana. Journal of Experimental Biology. 2001, 204: 4301–4309.
- ^ Staudacher, E.; Gebhardt, M.J.; Dürr, V. Antennal movements and mechanoreception: Neurobiology of active tactile sensors. Advances in Insect Physiology. 2005, 32: 49–205.
- ^ Servadei, A.; Zangheri, S.; Masutti, L. Entomologia generale ed applicata. CEDAM. 1972: 492–530.
- ^ Campbell, Frank L.; Priestly, June D. Flagellar Annuli of Blattella germanica (Dictyoptera: Blattellidae).–Changes in Their Numbers and Dimensions during Postembryonic Development. Annals of the Entomological Society of America. 1970, 63: 81–88. doi:10.1093/aesa/63.1.81.
- ^ Insect antennae. The Amateur Entomologists' Society. [2011-03-21].
- ^ 24.0 24.1 Insect Morphology. University of Minisotta (Department of Entomology). [2011-03-21]. (原始内容存档于2011-03-03).
- ^ Kirejtshuk, A.G. Head. Beetles (Coleoptera) and coleopterologist. zin.ru. November 2002 [2011-03-21].
- ^ 26.0 26.1 26.2 26.3 26.4 Gilliott, Cedric. Entomology 2. Springer-Verlag New York, LLC. August 1995. ISBN 0-306-44967-6.
- ^ Kapoor, V.C. C. Principles and Practices of Animal Taxonomy 1 1. Science Publishers. January 1998: 48. ISBN 1-57808-024-X.
- ^ 28.00 28.01 28.02 28.03 28.04 28.05 28.06 28.07 28.08 28.09 28.10 28.11 28.12 Chapman, R.F. The Insects: Structure and function 4th. Cambridge, New York: Cambridge University Press. 1998. ISBN 0-521-57048-4.
- ^ Mosquito biting mouthparts. allmosquitos.com. 2011 [April 17, 2011].
- ^ 30.0 30.1 30.2 30.3 Scoble, MJ. The Lepidoptera: Form, function, and diversity.. Oxford Univ. Press. 1992. ISBN 978-1-4020-6242-1.
- ^ 31.0 31.1 Evans, Arthur V.; Bellamy, Charles. An Inordinate Fondness for Beetles. April 2000. ISBN 0-520-22323-3.
- ^ Heppner, J. B. Butterflies and moths. Capinera, John L. (编). Encyclopedia of Entomology. Gale virtual reference library 4 2nd. Springer Reference. 2008: 4345. ISBN 978-1-4020-6242-1.
- ^ Agosta, Salvatore J.; Janzen, Daniel H. Body size distributions of large Costa Rican dry forest moths and the underlying relationship between plant and pollinator morphology. Oikos. 2004, 108 (1): 183–193. doi:10.1111/j.0030-1299.2005.13504.x.
- ^ Kunte, Krushnamegh. Allometry and functional constraints on proboscis lengths in butterflies (PDF). Functional Ecology. 2007, 21 (5): 982–987 [26 February 2011]. doi:10.1111/j.1365-2435.2007.01299.x. (原始内容 (PDF)存档于29 June 2011).
- ^ Sponging. University of Minissota. [April 17, 2011]. (原始内容存档于September 27, 2011).
- ^ Fly Mouthparts. School of Biological Sciences Online Learning Resources. University of Sydney. February 4, 2010 [April 17, 2011].
- ^ Meyer, John R. External Anatomy: WINGS. Department of Entomology, NC State University. 5 January 2007 [2011-03-21]. (原始内容存档于16 July 2011).
- ^ 38.00 38.01 38.02 38.03 38.04 38.05 38.06 38.07 38.08 38.09 38.10 38.11 38.12 38.13 38.14 38.15 38.16 38.17 38.18 38.19 Snodgrass, R. E. Principles of Insect Morphology. Cornell Univ Press. December 1993. ISBN 0-8014-8125-2.
- ^ Spieth, HT. A New Method of Studying the Wing Veins of the Mayflies and Some Results Therefrom (Ephemerida) (PDF). Entomological News. 1932. (原始内容 (PDF)存档于2011-09-30).
- ^ profemur, profemora - BugGuide.Net. bugguide.net. [10 December 2016].
- ^ protibia - BugGuide.Net. bugguide.net. [10 December 2016].
- ^ Schneiderman, Howard A. Discontinuous respiration in insects: role of the spiracles. Biol. Bull. 1960, 119 (3): 494–528. JSTOR 1539265. doi:10.2307/1539265.
- ^ Eisemann, WK; Jorgensen, W. K.; Merritt, D. J.; Rice, M. J.; Cribb, B. W.; Webb, P. D.; Zalucki, M. P.; et al. Do insects feel pain? — A biological view. Cellular and Molecular Life Sciences. 1984, 40 (2): 1420–1423. doi:10.1007/BF01963580.
- ^ Tracey, J; Wilson, RI; Laurent, G; Benzer, S; et al. painless, a Drosophila gene essential for nociception. Cell. 18 April 2003, 113 (2): 261–273. PMID 12705873. doi:10.1016/S0092-8674(03)00272-1.
- ^ Sømme, LS. Sentience and pain in invertebrates. Norwegian Scientific Committee for Food Safety. 14 January 2005 [September 30, 2009].[永久失效連結]
- ^ 46.0 46.1 General Entomology - Digestive and Excritory system. NC state University. [2009-05-03].
- ^ 47.0 47.1 47.2 Nation, James L. 15. Insect Physiology and Biochemistry 1. CRC Press. November 2001: 496pp. ISBN 0-8493-1181-0.
- ^ Duncan, Carl D. A Contribution to The Biology of North American Vespine Wasps 1. Stanford: Stanford University Press. 1939: 24–29.
- ^ Meyer, John R. Circulatory System. NC State University: Department of Entomology, NC State University: 1. 17 February 2006 [2009-10-11]. (原始内容存档于27 September 2009).
- ^ Chown, S.L.; S.W. Nicholson. Insect Physiological Ecology. New York: Oxford University Press. 2004. ISBN 0-19-851549-9.
- ^ Richard W. Merritt, Kenneth W. Cummins, and Martin B. Berg (editors). An Introduction to the Aquatic Insects of North America 4th. Kendall Hunt Publishers. 2007. ISBN 978-0-7575-5049-2.
- ^ Merritt, RW, KW Cummins, and MB Berg. An Introduction To The Aquatic Insects Of North America. Kendall Hunt Publishing Company. 2007. ISBN 0-7575-4128-3.
- ^ 53.0 53.1 McGavin, G. C. Essential Entomology; An order by order introduction. New York: Oxford University Press. 2001. ISBN 9780198500025.
- ^ 54.0 54.1 54.2 Triplehorn, C. A.; Johnson, N. F. Borror and DeLong's Introduction to the Study of Insects (7th). Brooks / Thomson Cole: Brooks / Thomson Cole. 2005.
- ^ 55.0 55.1 55.2 Elzinga, R.J. Fundamentals of Entomology 6th. New Jersey USA: Pearson/Prentice Hall. 2004.
- ^ Triplehorn, Charles A; Johnson, Norman F. Borror and DeLong's introduction to the study of insects. 7th. Australia: Thomson, Brooks/Cole. 2005. ISBN 9780030968358.
- ^ Gullan, P.J.; P.S. Cranston. The Insects: An Outline of Entomology 3. Oxford: Blackwell Publishing. 2005: 61–65. ISBN 1-4051-1113-5.
- ^ De Carlo; J. A. Hemipteros acuáticos y semiacuáticos. Estudio en grupos en las partes de igual función de los aparatos genitales masculinos de especies estudiadas. Revista de la Sociedad Entomológica Argentina. 1983, 42 (1/4): 149–154.
- ^ Andersen, N. Møller. Marine insects: genital morphology, phylogeny and evolution of sea skaters, genus Halobates (Hemiptera: Gerridae). Zoological Journal of the Linnean Society. 1991, 103 (1): 21–60. doi:10.1111/j.1096-3642.1991.tb00896.x.
- ^ Eggleton, P. Termites and trees: a review of recent advances in termite phylogenetics. Insectes Sociaux. 2001, 48: 187–193. doi:10.1007/pl00001766.
- ^ Lo, Nathan; Claudio Bandi; Hirofumi Watanabe; Christine Nalepa; Tiziana Beninat. Evidence for Cocladogenesis Between Diverse Dictyopteran Lineages and Their Intracellular Endosymbionts (PDF). Molecular Biology and Evolution. 2003, 20 (6): 907–913. PMID 12716997. doi:10.1093/molbev/msg097.
- ^ Leung, Chee Chee Leung. Cave may hold missing link. theage.com.au. March 22, 2007 [7 December 2013].
- ^ Lo, N; Beninati, T; Stone, F; Walker, J; Sacchi, L. Cockroaches that lack Blattabacterium endosymbionts: the phylogenetically divergent genus Nocticola. Biology Letters. 2007, 3 (3): 327–30. PMC 2464682
 . PMID 17376757. doi:10.1098/rsbl.2006.0614.
. PMID 17376757. doi:10.1098/rsbl.2006.0614.
- ^ Kunkel, Joseph G. How do cockroaches breathe?. The Cockroach FAQ. UMass Amherst. [7 December 2013].
- ^ 65.0 65.1 65.2 Gillot, C. Butterflies and moths. Entomology 2. 1995: 246–266 [14 November 2010]. ISBN 978-0-306-44967-3.
- ^ Schmidt-Nielsen, Knut. Insect Respiration. Animal Physiology: Adaptation and Environment 5 (illustrated). Cambridge University Press. Jan 15, 1997: 55 [6 March 2010]. ISBN 0-521-57098-0.
- ^ 67.0 67.1 67.2 Hoell, H.V., Doyen, J.T. & Purcell, A.H. Introduction to Insect Biology and Diversity, 2nd ed.. Oxford University Press. 1998: 493–499. ISBN 0-19-510033-6.
- ^ Williams, C. M. Physiology of insect diapause. II. Interaction between the pupal brain and prothoracic glands in the metamorphosis of the giant silkworm "Platysamia cecropia". Biol. Bull. 1947, 92: 89–180.
- ^ Lighton, J. R. B.; Lovegrove, B. G. A temperature-induced switch from diffusive to convective ventilation in the honeybee. Journal of Experimental Biology. 1990, 154: 509–516.










