鋅銅電池

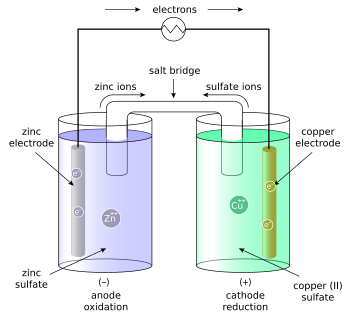

锌铜电池,又稱丹尼爾電池,是一种以锌为负電极;铜为正電极;硫酸鋅與硫酸銅為電解液的電化電池[1],由约翰·弗雷德里克·丹尼尔(John Frederic Daniell)於1836年發明,丹尼尔电池一稱來自其发明者的名字,丹尼爾為了消除伏打電堆中出現氫氣泡的問題,他的解決方法是使用第二種電解液來消耗只有一種電解液時會產生的氫氣。
熱力學理論[编辑]
在熱力學裏,電動勢 乘以電荷量 ,就是分離電荷所做的功項目。對於可逆過程,當電動勢促使電荷在電池內移動時,內能的變化包括這項目:
- ;
假設電池為丹尼爾電池,由於在這種電池內進行的反應不會產生氣體,系統體積不變,方程式簡化為
- 。
讓熵 為 和 的函數,熵的全微分為
- 。
假設等溫過程,那麼,方程式右手邊的第一個項目等於零:
- 。
將這方程式帶入內能的方程式:
- 。
這方程式右手邊的第二個項目是「充電熱」(heat of charging),定義為在一個等溫可逆的充電過程,系統的熱能吸收率 :
- 。
吸收率 比較不容易計算,可以找更有用的變數替換。思考亥姆霍茲自由能 :
- 。
所以, 是一對共軛變量(Conjugate variables)。其馬克士威關係式為:
- 。
帶入內能的方程式:
- 。
通常,電動勢跟溫度 、電荷量 有關。假若,能夠使丹尼爾電池內的溶液保持飽和狀態,有很多離子化合物隨時準備分解進入溶液,則電動勢跟電荷量無關,只跟溫度有關:
- 。
對於丹尼爾電池,體積不變,假設等壓過程,則焓的改變 ,稱為「反應熱」,等於內能的改變:
- 。
- ;
其中, 是金屬離子的電價, 是亞佛加厥常數, 是基本電荷量。
假設恆壓、恆體積,則電池的熱力學性質與電動勢的緊密關係,以方程式表達為
- 。
這樣,只要得到電動勢與溫度之間關係的資料,從測量電動勢和溫度的數據,很容易就能夠準確地計算出某化學反應的反應熱[3]。
参看[编辑]
参考文献[编辑]
- ^ Daniell Cell. [2017-03-22]. (原始内容存档于2021-04-20).
- ^ 可逆电池及其电动势的测定[永久失效連結]
- ^ Colin B P Finn. Thermal Physics. CRC Press. 1992: 163. ISBN 0748743790.
| |||||||||||||||||||||||||||||||||||





























