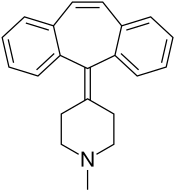
塞浦西他啶
| 此条目需要精通或熟悉医学的编者参与及协助编辑。 (2020年12月15日) |
 | |
 | |
| 临床资料 | |
|---|---|
| 读音 | (/ˌsaɪproʊˈhɛptədiːn/[1] |
| 商品名 | Periactin, others |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a682541 |
| 核准状况 | |
| 怀孕分级 |
|
| 给药途径 | Oral |
| ATC码 | |
| 法律规范状态 | |
| 法律规范 |
|
| 药物动力学数据 | |
| 血浆蛋白结合率 | 96 to 99% |
| 药物代谢 | Hepatic,[3][4] mostly CYP3A4 mediated. |
| 生物半衰期 | 8.6 hours[2] |
| 排泄途径 | 粪便 (2–20%; of which, 34% as unchanged drug) 与 肾 (40%; none as unchanged drug)[3][4] |
| 识别信息 | |
| |
| CAS号 | 129-03-3 969-33-5(盐酸盐) |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.004.482 |
| 化学信息 | |
| 化学式 | C21H21N |
| 摩尔质量 | 287.41 g·mol−1 |
| 3D模型(JSmol) | |
| |
| |
塞浦西他啶(Cyproheptadine),或名赛庚啶 ,是第一代抗组织胺药,具有抗乙酰胆碱和局部麻醉的功能,也是儿童抗过敏药水希普利敏的主要成分。
医疗使用[编辑]

- 塞浦西他啶用于治疗过敏反应 (特别是花粉症).[6] 用于此目的证实有效,但是第二代抗组织胺药像是酮替酚与氯雷他定也有相同的结果,且副作用较少。[7]
- 它也被用作偏头痛的预防性治疗。在2013年的一项研究中,开始治疗后7至10天内,患者的偏头痛发生率显著降低。这些患者在服药前偏头痛发作的平均频率为每月8.7次,开始治疗后3个月降至每月3.1次。[7][8] 在英国和其他一些国家/地区,药品仿单上有此用法。
- 它也被药品“仿单核准适应症外的使用”在婴儿周期性呕吐症候群的治疗。这种用途的唯一证据来自回顾性研究。[9]
- 塞浦西他啶有时被“仿单核准适应症外的使用”,以改善服用抗精神病药物的患者的静坐不能。[10]
- 它也被药品“仿单核准适应症外的使用”于治疗各种皮肤病,包括精神性搔痒症|[11]、药物诱发的多汗症(出汗过多)[12],以及预防表浅性单纯性水疱性表皮松解症的水疱形成。[13]
- 该药物的作用之一是食欲增加和体重增加,这导致该药物在消瘦的儿童以及囊肿性纤维化患者中用于此目的(在美国为“仿单核准适应症外的使用”)。[14][15][16]
- 它也使用在“仿单核准适应症外的使用”治疗中度至重度血清素综合症,与使用血清素类药物有关的复杂症状,像是选择性5-羟色胺再摄取抑制剂 (与单胺氧化酶抑制剂),以及在血清素产生的类癌肿瘤导致血液中5-羟色胺升高的案例。[17][18]
副作用[编辑]
- 镇静和困倦(通常是短暂的)Sedation and sleepiness (often transient)
- 头晕 Dizziness
- 协调不佳 Disturbed coordination
- 混乱 Confusion
- 躁动不安 Restlessness
- 兴奋 Excitation
- 紧张 Nervousness
- 震颤 Tremor
- 易怒 Irritability
- 失眠 Insomnia
- 感觉异常 Paresthesias
- 神经炎 Neuritis
- 抽搐 Convulsions
- 欣快感 Euphoria
- 幻觉 Hallucinations
- 歇斯底里 Hysteria
- 模糊 Faintness
- 皮疹和水肿的过敏表现 Allergic manifestation of rash and edema
- 发汗 Diaphoresis
- 荨麻疹 Urticaria
- 对光敏感 Photosensitivity
- 急性迷路炎 Acute labyrinthitis
- 复视(双眼) Diplopia (seeing double)
- 眩晕 Vertigo
- 耳鸣 Tinnitus
- 低血压 Hypotension (low blood pressure)
- 心悸 Palpitation
- 期前收缩 Extrasystoles
- 过敏性休克 Anaphylactic shock
- 溶血性贫血 Hemolytic anemia
- 诸如白细胞减少症,粒细胞缺乏症和血小板减少症等血液异常 Blood dyscrasias such as leukopenia, agranulocytosis and thrombocytopenia
- 胆汁淤积 Cholestasis
- 对肝脏的影响:
- 上腹窘迫 Epigastric distress
- 食欲不振 Anorexia
- 恶心 Nausea
- 呕吐 Vomiting
- 腹泻 Diarrhea
- 抗胆碱能副作用:
- 视力模糊 Blurred vision
- 便秘 Constipation
- 口腔干燥症(口干) Xerostomia (dry mouth)
- 心动过速(高心率)Tachycardia (high heart rate)
- 尿潴留 Urinary retention
- 排尿困难 Difficulty passing urine
- 鼻塞 Nasal congestion
- 鼻或喉咙干燥 Nasal or throat dryness
- 频尿 Urinary frequency
- 早期月经 Early menses
- 支气管分泌物增厚 Thickening of bronchial secretions
- 胸闷气喘 Tightness of chest and wheezing
- 疲劳 Fatigue
- 寒意 Chills
- 头痛 Headache
- 食欲增加 Increased appetite
- 体重增加 Weight gain
用药过量[编辑]
过量使用时,有时建议使用活性炭进行洗胃。这些症状通常表明中枢神经系统抑制(或在某些情况下相反地刺激中枢神经系统)和过度的抗胆碱能副作用。小鼠的半数致死量(LD50)为 123 mg/kg,,大鼠的半数致死量为 295 mg/kg 。[3][4]
药理[编辑]
药效学[编辑]
塞浦西他啶对此表列出的所有受体均表现为拮抗剂或逆向激动剂。[20]
| Site | Ki (nM)[a] | Action[b] | Species | Ref. |
|---|---|---|---|---|
| H1 | 0.06 | ↓ | Human | |
| H2 | ND | ND | ||
| H3 | >10,000 | Human | ||
| H4 | 202 | Human | ||
| M1 | 12 | ↓ | Human | |
| M2 | 7 | ↓ | Human | |
| M3 | 12 | ↓ | Human | |
| M4 | 8 | ↓ | Human | |
| M5 | 11.8 | ↓ | Human | |
| 5-HT1A | 59 | Human | ||
| 5-HT2A | 1.67 | ↓ | Human | |
| 5-HT2B | 1.54 | ↓ | Human | |
| 5-HT2C | 2.23 | ↓ | Human | |
| 5-HT3 | 228 | Mouse | ||
| 5-HT6 | 142 | Human | ||
| 5-HT7 | 123 | Human | ||
| D1 | 117 | Human | ||
| D2 | 112 | ↓ | Human | |
| D3 | 8 | Human | ||
| SERT | 4,100 | Rat | ||
| NET | 290 | Rat | ||
| DAT | ND | ND | ||
塞浦西他啶是一种非常有效的抗组胺药或H1受体的拮抗剂。它在较高浓度下还具有抗胆碱能、抗血清素能和抗多巴胺能活性。它是5-HT2受体的强效拮抗剂,这是其治疗血清素综合症的基础。
药物代谢动力学[编辑]
塞浦西他啶口服后吸收良好,血浆浓度峰值出现在1至3小时后。[22]口服塞浦西他啶的生物半衰期约为8小时。[2]
化学[编辑]
塞浦西他啶是三环苯并环庚烯,与吡唑替芬和酮替酚以及三环抗抑郁药密切相关。
研究[编辑]
在一项规模较小的精神分裂症患者中,辅助使用塞浦西他啶作为辅助治疗,该患者的病情稳定且正在接受其他药物治疗。虽然注意力和口语流利性似乎有所改善,但这项研究规模太小,不足以概括。[23]在另两项针对精神分裂症患者的试验中,也已对其进行了佐剂研究,总共约有50人,并且似乎没有效果。[24]
已经进行了一些试验,以观察塞浦西他啶是否可以减轻SSRI和抗精神病药引起的性功能障碍。[25]
塞浦西他啶已被研究用于创伤后压力症候群(PTSD)。[24]
兽医用途[编辑]
塞浦西他啶使用在猫的食欲刺激剂[26] ,也可作为哮喘的辅助治疗剂。[27] 可能的副作用包括刺激和攻击行为。[28]它在猫的生物半衰期为 12 小时。[27]
塞浦西他啶也用在马的垂体中间部功能障碍的二线治疗。[29][30]
参考资料[编辑]
- ^ Cyproheptadine. Dictionary.com Unabridged. Random House.
- ^ 2.0 2.1 Gunja N, Collins M, Graudins A. A comparison of the pharmacokinetics of oral and sublingual cyproheptadine. Journal of Toxicology. Clinical Toxicology. 2004, 42 (1): 79–83. PMID 15083941. S2CID 20196551. doi:10.1081/clt-120028749.
- ^ 3.0 3.1 3.2 3.3 CYPROHEPTADINE HYDROCHLORIDE tablet [Boscogen, Inc.]. DailyMed. Boscogen, Inc. November 2010 [26 October 2013]. (原始内容 (PDF)存档于2013-07-04).
- ^ 4.0 4.1 4.2 4.3 PRODUCT INFORMATION PERIACTIN® (cyproheptadine hydrochloride) (PDF). Aspen Pharmacare Australia. Aspen Pharmacare Australia Pty Ltd. 17 November 2011 [26 October 2013]. (原始内容 (PDF)存档于29 October 2013).
- ^ Fischer, Jnos; Ganellin, C. Robin. Analogue-based Drug Discovery. John Wiley & Sons. 2006: 547 [2020-12-15]. ISBN 9783527607495. (原始内容存档于2021-08-29) (英语).
- ^ MedlinePlus Drug Information: Cyproheptadine. [2020-12-15]. (原始内容存档于2008-10-01).
- ^ 7.0 7.1 De Bruyne, P; Christiaens, T; Boussery, K; Mehuys, E; Van Winckel, M. Are antihistamines effective in children? A review of the evidence. Archives of Disease in Childhood. January 2017, 102 (1): 56–60. PMID 27335428. S2CID 21185048. doi:10.1136/archdischild-2015-310416.
- ^ Saito, Y; Yamanaka, G; Shimomura, H; Shiraishi, K; Nakazawa, T; Kato, F; Shimizu-Motohashi, Y; Sasaki, M; Maegaki, Y. Reconsideration of the diagnosis and treatment of childhood migraine: A practical review of clinical experiences. Brain & Development. May 2017, 39 (5): 386–394. PMID 27993427. S2CID 34703034. doi:10.1016/j.braindev.2016.11.011.
- ^ Salvatore, S; Barberi, S; Borrelli, O; Castellazzi, A; Di Mauro, D; Di Mauro, G; Doria, M; Francavilla, R; Landi, M; Martelli, A; Miniello, VL; Simeone, G; Verduci, E; Verga, C; Zanetti, MA; Staiano, A; SIPPS Working Group on, FGIDs. Pharmacological interventions on early functional gastrointestinal disorders. Italian Journal of Pediatrics. 16 July 2016, 42 (1): 68. PMC 4947301
 . PMID 27423188. doi:10.1186/s13052-016-0272-5.
. PMID 27423188. doi:10.1186/s13052-016-0272-5.
- ^ Taylor, David; Paton, Carol; Kapur, Shitij. The Maudsley Prescribing Guidelines in Psychiatry. John Wiley & Sons. 2015: 85 [2020-12-15]. ISBN 9781118754573. (原始内容存档于2021-08-28) (英语).
- ^ Szepietowski, JC; Reszke, R. Psychogenic Itch Management 50. 2016: 124–32. ISBN 978-3-318-05888-8. PMID 27578081. doi:10.1159/000446055.
|journal=被忽略 (帮助) - ^ Ashton AK, Weinstein WL. Cyproheptadine for drug-induced sweating. American Journal of Psychiatry. May 2002, 159 (5): 874–5 [2020-12-15]. PMID 11986151. doi:10.1176/appi.ajp.159.5.874-a. (原始内容存档于2012-03-19).
- ^ Pfendner, Ellen G.; Bruckner, Anna L. Epidermolysis Bullosa Simplex. GeneReviews. October 13, 2016 [2020-12-15]. PMID 20301543. (原始内容存档于2020-10-22).
- ^ Ciproheptadina, estimulante del apetito (Cyproheptadine, appetite stimulant). [2020-12-15]. (原始内容存档于2020-10-24).
- ^ Bioplex NF. [2020-12-15]. (原始内容存档于2018-04-18).
- ^ Harrison ME, Norris ML, Robinson A, Spettigue W, Morrissey M, Isserlin L. Use of cyproheptadine to stimulate appetite and body weight gain: A systematic review.. Appetite. 2019, 137: 62–72. PMID 30825493. S2CID 72333631. doi:10.1016/j.appet.2019.02.012.
- ^ Rossi, S (编). Australian Medicines Handbook 2013. Adelaide: The Australian Medicines Handbook Unit Trust. 2013. ISBN 978-0-9805790-9-3.
- ^ Iqbal, MM; Basil, MJ; Kaplan, J; Iqbal, MT. Overview of serotonin syndrome. Annals of Clinical Psychiatry. November 2012, 24 (4): 310–8. PMID 23145389.
- ^ Chertoff, Jason. Cyproheptadine-Induced Acute Liver Failure. ACG Case Reports Journal. 8 July 2014, 1 (4): 212–213. PMC 4286888
 . PMID 25580444. doi:10.14309/crj.2014.56.
. PMID 25580444. doi:10.14309/crj.2014.56.
- ^ 20.0 20.1 Roth, BL; Driscol, J. PDSP Ki Database. Psychoactive Drug Screening Program (PDSP). University of North Carolina at Chapel Hill and the United States National Institute of Mental Health. [14 August 2017]. (原始内容存档于2021-08-28).
- ^ Pucci E, Petraglia F. Treatment of androgen excess in females: yesterday, today and tomorrow. Gynecol. Endocrinol. December 1997, 11 (6): 411–33. PMID 9476091. doi:10.3109/09513599709152569.
- ^ Lindsay Murray; Frank Daly; David McCoubrie; Mike Cadogan. Toxicology Handbook. Elsevier Australia. 15 January 2011: 388 [27 November 2011]. ISBN 978-0-7295-3939-5. (原始内容存档于2014-01-01).
- ^ Buoli, M; Altamura, AC. May non-antipsychotic drugs improve cognition of schizophrenia patients?. Pharmacopsychiatry. March 2015, 48 (2): 41–50. PMID 25584772. doi:10.1055/s-0034-1396801.
- ^ 24.0 24.1 Dabaghzadeh, F; Khalili, H; Ghaeli, P; Dashti-Khavidaki, S. Potential benefits of cyproheptadine in HIV-positive patients under treatment with antiretroviral drugs including efavirenz. Expert Opinion on Pharmacotherapy. December 2012, 13 (18): 2613–24. PMID 23140169. S2CID 25769557. doi:10.1517/14656566.2012.742887.
- ^ Nunes, LV; Moreira, HC; Razzouk, D; Nunes, SO; Mari Jde, J. Strategies for the treatment of antipsychotic-induced sexual dysfunction and/or hyperprolactinemia among patients of the schizophrenia spectrum: a review. Journal of Sex & Marital Therapy. 2012, 38 (3): 281–301. PMID 22533871. S2CID 23406005. doi:10.1080/0092623X.2011.606883.
- ^ Agnew, W; Korman, R. Pharmacological appetite stimulation: rational choices in the inappetent cat. Journal of Feline Medicine and Surgery. September 2014, 16 (9): 749–56. PMID 25146662. S2CID 37126352. doi:10.1177/1098612X14545273.
- ^ 27.0 27.1 Dowling PM. Systemic Therapy of Airway Disease: Cyproheptadine. Kahn CM, Line S, Aiello SE (编). The Merck Veterinary Manual 9th. John Wiley & Sons. February 8, 2005 [2022-06-16]. ISBN 978-0-911910-50-6. (原始内容存档于2016-03-04). Retrieved on October 26, 2008.
- ^ Dowling PM. Drugs Affecting Appetite. Kahn CM, Line S, Aiello SE (编). The Merck Veterinary Manual 9th. John Wiley & Sons. February 8, 2005 [2022-06-16]. ISBN 978-0-911910-50-6. (原始内容存档于2012-10-28). Retrieved on October 26, 2008.
- ^ Durham, AE. Therapeutics for Equine Endocrine Disorders. The Veterinary Clinics of North America. Equine Practice. April 2017, 33 (1): 127–139. PMID 28190613. doi:10.1016/j.cveq.2016.11.003.
- ^ Merck Vet Manual. Hirsutism Associated with Adenomas of the Pars Intermedia. [April 24, 2011]. (原始内容存档于2016-03-03).
| ||||||||||||||||||||||||||||||||||||||||||
