血管紧张素转化酶2:修订间差异
小无编辑摘要 |
小 →冠狀病毒受體 |
||
| 第36行: | 第36行: | ||
==冠狀病毒受體== |
==冠狀病毒受體== |
||
血管紧张素转化酶2被許多[[冠狀病毒]]用來當作感染細胞的受體,包括造成[[普通感冒]]的[[人類冠狀病毒NL63]]<ref name="NCBI_ACE2">{{cite web|title=Gene: ACE2, angiotensin I converting enzyme 2 |work=[[National Center for Biotechnology Information]] (NCBI) |publisher=U.S. National Library of Medicine | date=2020-02-28 | url=https://www.ncbi.nlm.nih.gov/gene/59272 }}</ref>、造成[[SARS]]的[[SARS病毒]]<ref name="Fehr_2015">{{cite book | vauthors = Fehr AR, Perlman S | title = Coronaviruses | chapter = Coronaviruses: an overview of their replication and pathogenesis | series = Methods in Molecular Biology | volume = 1282 | pages = 1–23 | year = 2015 | pmid = 25720466 | pmc = 4369385 | doi = 10.1007/978-1-4939-2438-7_1 | publisher = Springer New York | isbn = 978-1-4939-2437-0 | quote = Many α-coronaviruses utilize aminopeptidase N (APN) as their receptor, SARS-CoV and HCoV-NL63 use angiotensin-converting enzyme 2 (ACE2) as their receptor, MHV enters through CEACAM1, and the recently identified MERS-CoV binds to dipeptidyl-peptidase 4 (DPP4) to gain entry into human cells (See Table 1 for a list of known CoV receptors). }}</ref><ref>{{cite journal | vauthors = Li F | title = Receptor recognition and cross-species infections of SARS coronavirus | journal = Antiviral Research | volume = 100 | issue = 1 | pages = 246–54 | date = October 2013 | pmid = 23994189 | pmc = 3840050 | doi = 10.1016/j.antiviral.2013.08.014 }}</ref>和造成[[2019冠狀病毒病]]的[[SARS-CoV-2]]<ref>{{cite web |title=What are the official names of the disease and the virus that causes it? |url=https://www.who.int/news-room/q-a-detail/q-a-coronaviruses |website=Q&A on coronaviruses |publisher=World Health Organization |access-date=22 February 2020 |archive-url=https://web.archive.org/web/20200305015146/https://www.who.int/news-room/q-a-detail/q-a-coronaviruses |archive-date=5 March 2020}}</ref>,這些病毒刺突蛋白S1結構域中的[[受體結合結構域]](receptor binding domain;RBD)可和ACE2胞外的區域結合後,刺突蛋白可能被細胞表面的[[跨膜丝氨酸蛋白酶2]](TMPRSS2)切割,促使病毒外膜和宿主[[細胞膜]]融合而讓病毒進入[[細胞質]]<ref>{{Cite journal|last1=Akhmerov Akbarshakh|last2=Marban Eduardo|title=COVID-19 and the Heart|journal=Circulation Research|year=2020|volume=0|issue=10|pages=1443–1455|doi=10.1161/CIRCRESAHA.120.317055|pmid=32252591|pmc=7188058}}</ref>;此外SARS-CoV與SARS-CoV-2<ref name="pmid32221306">{{cite journal| author=Ou X, Liu Y, Lei X, Li P, Mi D, Ren L | display-authors=etal| title=Characterization of spike glycoprotein of SARS-CoV-2 on virus entry and its immune cross-reactivity with SARS-CoV. | journal=Nat Commun | year= 2020 | volume= 11 | issue= 1 | pages= 1620 | pmid=32221306 | doi=10.1038/s41467-020-15562-9 | pmc=7100515 | url=https://www.ncbi.nlm.nih.gov/entrez/eutils/elink.fcgi?dbfrom=pubmed&tool=sumsearch.org/cite&retmode=ref&cmd=prlinks&id=32221306 }} </ref>還可能在不被TMPRSS2切割的情況下,與ACE2受體一起藉由[[內吞作用]]進入細胞,隨後其刺突蛋白在[[溶體]]中被{{le|組織蛋白酶|Cathepsin}}切割後,再從溶體進入細胞質中<ref name="Wang_2008">{{cite journal | vauthors = Wang H, Yang P, Liu K, Guo F, Zhang Y, Zhang G, Jiang C | title = SARS coronavirus entry into host cells through a novel clathrin- and caveolae-independent endocytic pathway | journal = Cell Research | volume = 18 | issue = 2 | pages = 290–301 | date = February 2008 | pmid = 18227861 | pmc = 7091891 | doi = 10.1038/cr.2008.15 }}</ref><ref name="Millet_2018">{{cite journal | vauthors = Millet JK, Whittaker GR | title = Physiological and molecular triggers for SARS-CoV membrane fusion and entry into host cells | journal = Virology | volume = 517 | pages = 3–8 | date = April 2018 | pmid = 29275820 | pmc = 7112017 | doi = 10.1016/j.virol.2017.12.015 }}</ref><ref name="pmid16339146">{{cite journal| author=Huang IC, Bosch BJ, Li F, Li W, Lee KH, Ghiran S | display-authors=etal| title=SARS coronavirus, but not human coronavirus NL63, utilizes cathepsin L to infect ACE2-expressing cells. | journal=J Biol Chem | year= 2006 | volume= 281 | issue= 6 | pages= 3198-203 | pmid=16339146 | doi=10.1074/jbc.M508381200 | pmc= | url=https://www.ncbi.nlm.nih.gov/entrez/eutils/elink.fcgi?dbfrom=pubmed&tool=sumsearch.org/cite&retmode=ref&cmd=prlinks&id=16339146}}</ref>。 |
血管紧张素转化酶2被許多[[冠狀病毒]]用來當作感染細胞的受體,包括造成[[普通感冒]]的[[人類冠狀病毒NL63]]<ref name="NCBI_ACE2">{{cite web|title=Gene: ACE2, angiotensin I converting enzyme 2 |work=[[National Center for Biotechnology Information]] (NCBI) |publisher=U.S. National Library of Medicine | date=2020-02-28 | url=https://www.ncbi.nlm.nih.gov/gene/59272 }}</ref>、造成[[SARS]]的[[SARS病毒]]<ref name="Fehr_2015">{{cite book | vauthors = Fehr AR, Perlman S | title = Coronaviruses | chapter = Coronaviruses: an overview of their replication and pathogenesis | series = Methods in Molecular Biology | volume = 1282 | pages = 1–23 | year = 2015 | pmid = 25720466 | pmc = 4369385 | doi = 10.1007/978-1-4939-2438-7_1 | publisher = Springer New York | isbn = 978-1-4939-2437-0 | quote = Many α-coronaviruses utilize aminopeptidase N (APN) as their receptor, SARS-CoV and HCoV-NL63 use angiotensin-converting enzyme 2 (ACE2) as their receptor, MHV enters through CEACAM1, and the recently identified MERS-CoV binds to dipeptidyl-peptidase 4 (DPP4) to gain entry into human cells (See Table 1 for a list of known CoV receptors). }}</ref><ref>{{cite journal | vauthors = Li F | title = Receptor recognition and cross-species infections of SARS coronavirus | journal = Antiviral Research | volume = 100 | issue = 1 | pages = 246–54 | date = October 2013 | pmid = 23994189 | pmc = 3840050 | doi = 10.1016/j.antiviral.2013.08.014 }}</ref>和造成[[2019冠狀病毒病]]的[[SARS-CoV-2]]<ref>{{cite web |title=What are the official names of the disease and the virus that causes it? |url=https://www.who.int/news-room/q-a-detail/q-a-coronaviruses |website=Q&A on coronaviruses |publisher=World Health Organization |access-date=22 February 2020 |archive-url=https://web.archive.org/web/20200305015146/https://www.who.int/news-room/q-a-detail/q-a-coronaviruses |archive-date=5 March 2020}}</ref>,這些病毒刺突蛋白S1結構域中的[[受體結合結構域]](receptor binding domain;RBD)可和ACE2胞外的區域結合後,刺突蛋白可能被細胞表面的[[跨膜丝氨酸蛋白酶2]](TMPRSS2)切割,促使病毒外膜和宿主[[細胞膜]]融合而讓病毒進入[[細胞質]]<ref>{{Cite journal|last1=Akhmerov Akbarshakh|last2=Marban Eduardo|title=COVID-19 and the Heart|journal=Circulation Research|year=2020|volume=0|issue=10|pages=1443–1455|doi=10.1161/CIRCRESAHA.120.317055|pmid=32252591|pmc=7188058}}</ref>;此外SARS-CoV與SARS-CoV-2<ref name="pmid32221306">{{cite journal| author=Ou X, Liu Y, Lei X, Li P, Mi D, Ren L | display-authors=etal| title=Characterization of spike glycoprotein of SARS-CoV-2 on virus entry and its immune cross-reactivity with SARS-CoV. | journal=Nat Commun | year= 2020 | volume= 11 | issue= 1 | pages= 1620 | pmid=32221306 | doi=10.1038/s41467-020-15562-9 | pmc=7100515 | url=https://www.ncbi.nlm.nih.gov/entrez/eutils/elink.fcgi?dbfrom=pubmed&tool=sumsearch.org/cite&retmode=ref&cmd=prlinks&id=32221306 }} </ref>還可能在不被TMPRSS2切割的情況下,與ACE2受體一起藉由[[內吞作用]]進入細胞,隨後其刺突蛋白在[[溶體]]中被{{le|組織蛋白酶|Cathepsin}}切割後,再從溶體進入細胞質中<ref name="Wang_2008">{{cite journal | vauthors = Wang H, Yang P, Liu K, Guo F, Zhang Y, Zhang G, Jiang C | title = SARS coronavirus entry into host cells through a novel clathrin- and caveolae-independent endocytic pathway | journal = Cell Research | volume = 18 | issue = 2 | pages = 290–301 | date = February 2008 | pmid = 18227861 | pmc = 7091891 | doi = 10.1038/cr.2008.15 }}</ref><ref name="Millet_2018">{{cite journal | vauthors = Millet JK, Whittaker GR | title = Physiological and molecular triggers for SARS-CoV membrane fusion and entry into host cells | journal = Virology | volume = 517 | pages = 3–8 | date = April 2018 | pmid = 29275820 | pmc = 7112017 | doi = 10.1016/j.virol.2017.12.015 }}</ref><ref name="pmid16339146">{{cite journal| author=Huang IC, Bosch BJ, Li F, Li W, Lee KH, Ghiran S | display-authors=etal| title=SARS coronavirus, but not human coronavirus NL63, utilizes cathepsin L to infect ACE2-expressing cells. | journal=J Biol Chem | year= 2006 | volume= 281 | issue= 6 | pages= 3198-203 | pmid=16339146 | doi=10.1074/jbc.M508381200 | pmc= | url=https://www.ncbi.nlm.nih.gov/entrez/eutils/elink.fcgi?dbfrom=pubmed&tool=sumsearch.org/cite&retmode=ref&cmd=prlinks&id=16339146}}</ref>。 |
||
SARS-CoV-2刺突蛋白的受體結構域有6個[[胺基酸]]為與宿主細胞的ACE2結合所需,包括[[白胺酸]]455、[[苯丙胺酸]]486、[[麩醯胺酸]]493、[[絲胺酸]]494、[[天門冬醯胺]]501與[[酪氨酸]]505<ref name="pmid32284615">{{cite journal| author=Andersen KG, Rambaut A, Lipkin WI, Holmes EC, Garry RF| title=The proximal origin of SARS-CoV-2. | journal=Nat Med | year= 2020 | volume= 26 | issue= 4 | pages= 450-452 | pmid=32284615 | doi=10.1038/s41591-020-0820-9 | pmc=7095063 | url=https://www.ncbi.nlm.nih.gov/entrez/eutils/elink.fcgi?dbfrom=pubmed&tool=sumsearch.org/cite&retmode=ref&cmd=prlinks&id=32284615 }} </ref>。 |
|||
==註腳== |
==註腳== |
||
2021年3月3日 (三) 00:28的版本
血管紧张素转化酶2(英語:Angiotensin-converting enzyme 2;ACE2,人類的ACE2常被稱為hACE2[5])在人類基因組中由X染色體上的基因編碼,是一種表現於肺、動脈、心臟、腎臟與腸道等組織細胞表面的膜蛋白,可分別將血管紧张素I和血管紧张素II轉化为血管收縮素(1-7)和血管收縮素(1-9)[6][7]。此外,ACE2还被SARS-CoV、SARS-CoV-2和人類冠狀病毒NL63等冠狀病毒用作感染細胞的受体[8]。
結構
| 血管紧张素转化酶2 | |||||||
|---|---|---|---|---|---|---|---|
| |||||||
| 识别码 | |||||||
| EC編號 | 3.4.17.23 | ||||||
| 数据库 | |||||||
| IntEnz | IntEnz浏览 | ||||||
| BRENDA | BRENDA入口 | ||||||
| ExPASy | NiceZyme浏览 | ||||||
| KEGG | KEGG入口 | ||||||
| MetaCyc | 代谢路径 | ||||||
| PRIAM | 概述 | ||||||
| PDB | RCSB PDB PDBj PDBe PDBsum | ||||||
| |||||||
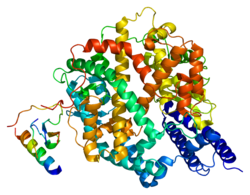
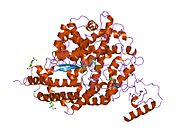
血管紧张素转化酶2(ACE2)最早於2000年自cDNA基因庫中被發現,為血管紧张素转化酶(ACE)第一個被發現的旁系同源體[6],ACE2的基因位於人類基因組中的X染色體,包括18個外顯子,編碼的蛋白由805個胺基酸組成,與ACE的胺基酸序列相似度為42%[9],是一個帶有鋅離子的金屬蛋白,屬單次跨膜蛋白(第一型膜蛋白),其N端結構域為一M2多肽酶,位於細胞膜外側;C端則與另一種名為collectrin的蛋白同源,包括疏水的跨膜結構域和一個胺基酸轉運體結構域,位於細胞內[10]。
ACE2因有跨膜區域而造成其結構測定的困難,過去僅知其N端多肽酶的結構,直到2020年科學家才用低溫電子顯微鏡測出了與另一蛋白B0AT1結合狀態的完整ACE2結構,發現兩個ACE2和兩個B0AT1組成一複合體,複合體中兩個ACE2有交互作用,B0AT1間則無交互作用,僅與鄰近的ACE2作用,因此研究人員推測細胞膜上的ACE2也可能會形成二聚體[11][12]。
表現組織
人體幾乎所有器官組織都有表現血管紧张素转化酶(ACE),而血管紧张素转化酶2則表現於II型肺泡細胞、小腸腸上皮細胞、血管內皮細胞、血管平滑肌細胞、腎臟上皮細胞等,腦部許多神經元與膠細胞可能也有表現ACE2[9][13]。
功能
切割多肽
血管紧张素转化酶2主要的功能是與血管紧张素Ⅰ转化酶(ACE)拮抗,ACE可將無活性的血管收縮素Ⅰ切割成血管收縮素II,後者可促進抗利尿激素與醛固酮的分泌,以及刺激血管平滑肌收縮,使血壓上升;ACE2則分解血管收縮素Ⅰ和血管收縮素II以抑制其作用,將其C端的胺基酸移除,分別將前者轉化成血管收縮素(1-9),將後者轉化成血管收縮素(1-7)[註 1],其中切割血管收縮素II比切割血管收縮素I的能力高出許多,切割的產物中,血管收縮素(1-9)的功能不明,血管收縮素(1-7)則可刺激一氧化氮合成、抑制MAPK/ERK途徑、抑制活性氧類的生成以及抑制TGFβ途徑,因此在心血管組織中有抗氧化與抗發炎等功能[9][11]。許多研究結果顯示ACE2表現量的下降與數種心血管疾病有相關性[15]。
肺泡細胞表現的ACE2有保護肺組織的功能。血管收縮素II可促進肺泡細胞凋亡與肺纖維化[16],因此ACE2將前者分解成也會將血管收縮素II切割成血管收縮素(1-7),可保護肺免於損傷[17],加上血管收縮素(1-7)可與MAS1受體結合,啟動下游反應以抑制血管收縮素II的作用[17][18]。
在骨骼肌中,血管收縮素II與血管收縮素(1-7)均有重要功能。血管收縮素II透過多種途徑降低肌肉蛋白質的合成,包括抑制Akt-mTOR途徑、促進肌萎缩素1與肌环指蛋白1的合成、生成活性氧物質而活化胱天蛋白酶途徑使細胞凋亡等,肌肉蛋白合成與分解的失衡會造成肌萎缩、肌纖維化等症狀[19][20]。將血管收縮素II轉化成血管收縮素(1-7)可停止其作用,且後者還可與MAS1受體結合,活化另一條反應途徑而抑制肌纖維化[11][21]。相較之下ACE2在骨骼肌的直接影響還有待更多研究闡明,有初步研究結果顯示在萎縮的肌肉組織中,ACE2可能可降低纖維化[11][22]。
其他
除了切割多肽外,ACE2還有些與其蛋白酶活性無關的功能。有研究顯示ACE2可與整合素結合,有助於細胞黏附[11][23]。此外ACE2還參與了另一蛋白B0AT1的膜囊泡運輸過程,為其伴護蛋白,與B0AT1形成一複合體,協助將其轉運至細胞膜上[12]。
移除
ACE2的跨膜結構域可被一種稱為金屬蛋白酶17(MMP17)的脱落酶切割,將其胞外部分釋放到血液中,進而從組織間移除[24][25],此過程受到許多調控,例如有一種鈣調蛋白可與ACE2結合以抑制MMP17的切割[26],血管收縮素II也可促進MMP17的活性,把會將其分解的ACE2移除[27],另外許多病理狀況、發炎反應也可促進MMP17對ACE2的切割。脫落酶的切割會造成心血管組織中ACE2的流失、血液中ACE2的濃度升高,因此後者可當作心臟衰竭、心房顫動、動脈粥樣硬化、慢性腎臟病、心肌梗塞與中風等多種疾病的生物標記[11][28]。
冠狀病毒受體
血管紧张素转化酶2被許多冠狀病毒用來當作感染細胞的受體,包括造成普通感冒的人類冠狀病毒NL63[29]、造成SARS的SARS病毒[30][31]和造成2019冠狀病毒病的SARS-CoV-2[32],這些病毒刺突蛋白S1結構域中的受體結合結構域(receptor binding domain;RBD)可和ACE2胞外的區域結合後,刺突蛋白可能被細胞表面的跨膜丝氨酸蛋白酶2(TMPRSS2)切割,促使病毒外膜和宿主細胞膜融合而讓病毒進入細胞質[33];此外SARS-CoV與SARS-CoV-2[34]還可能在不被TMPRSS2切割的情況下,與ACE2受體一起藉由內吞作用進入細胞,隨後其刺突蛋白在溶體中被組織蛋白酶切割後,再從溶體進入細胞質中[35][36][37]。
SARS-CoV-2刺突蛋白的受體結構域有6個胺基酸為與宿主細胞的ACE2結合所需,包括白胺酸455、苯丙胺酸486、麩醯胺酸493、絲胺酸494、天門冬醯胺501與酪氨酸505[38]。
註腳
参考文献
- ^ 1.0 1.1 1.2 GRCh38: Ensembl release 89: ENSG00000130234 - Ensembl, May 2017
- ^ 2.0 2.1 2.2 GRCm38: Ensembl release 89: ENSMUSG00000015405 - Ensembl, May 2017
- ^ Human PubMed Reference:. National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ Mouse PubMed Reference:. National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ Kasmi Y, Khataby K, Souiri A. Coronaviridae: 100,000 Years of Emergence and Reemergence. Ennaji MM (编). Emerging and Reemerging Viral Pathogens. Volume 1: Fundamental and Basic Virology Aspects of Human, Animal and Plant Pathogens. Elsevier. 2019: 135. ISBN 978-0-12-819400-3.
- ^ 6.0 6.1 Donoghue M, Hsieh F, Baronas E, Godbout K, Gosselin M, Stagliano N; et al. A novel angiotensin-converting enzyme-related carboxypeptidase (ACE2) converts angiotensin I to angiotensin 1-9.. Circ Res. 2000, 87 (5): E1–9. PMID 10969042. doi:10.1161/01.res.87.5.e1.
- ^ Keidar S, Kaplan M, Gamliel-Lazarovich A. ACE2 of the heart: From angiotensin I to angiotensin (1-7). Cardiovasc Res. 2007, 73 (3): 463–9. PMID 17049503.
- ^ Weiss SR, Navas-Martin S. Angiotensin-converting enzyme 2--a new cardiac regulator. Microbiol Mol Biol Rev. 2005, 69 (4): 635–64. PMID 16339739.
- ^ 9.0 9.1 9.2 Burrell LM, Johnston CI, Tikellis C, Cooper ME. ACE2, a new regulator of the renin-angiotensin system.. Trends Endocrinol Metab. 2004, 15 (4): 166–9. PMC 7128798
 . PMID 15109615. doi:10.1016/j.tem.2004.03.001.
. PMID 15109615. doi:10.1016/j.tem.2004.03.001.
- ^ Turner AJ. Chapter 25: ACE2 Cell Biology, Regulation, and Physiological Functions. Unger T, Ulrike M, Steckelings UM, dos Santos RA (编). The Protective Arm of the Renin Angiotensin System (RAS): Functional Aspects and Therapeutic Implications. Academic Press. 2015: 185–189. ISBN 978-0-12-801364-9. doi:10.1016/B978-0-12-801364-9.00025-0.
- ^ 11.0 11.1 11.2 11.3 11.4 11.5 11.6 Yamamoto K, Takeshita H, Rakugi H. ACE2, angiotensin 1-7 and skeletal muscle: review in the era of COVID-19.. Clin Sci (Lond). 2020, 134 (22): 3047–3062. PMC 7687025
 . PMID 33231620. doi:10.1042/CS20200486.
. PMID 33231620. doi:10.1042/CS20200486.
- ^ 12.0 12.1 Yan R, Zhang Y, Li Y, Xia L, Guo Y, Zhou Q. Structural basis for the recognition of SARS-CoV-2 by full-length human ACE2.. Science. 2020, 367 (6485): 1444–1448. PMC 7164635
 . PMID 32132184. doi:10.1126/science.abb2762.
. PMID 32132184. doi:10.1126/science.abb2762.
- ^ Kabbani, Nadine; Olds, James L. Does COVID19 infect the brain? If so, smokers might be at a higher risk. Molecular Pharmacology. 1 April 2020, 97 (5): 351–353. PMC 7237865
 . PMID 32238438. doi:10.1124/molpharm.120.000014.
. PMID 32238438. doi:10.1124/molpharm.120.000014.
- ^ Domenig O, Manzel A, Grobe N, Königshausen E, Kaltenecker CC, Kovarik JJ; et al. Neprilysin is a Mediator of Alternative Renin-Angiotensin-System Activation in the Murine and Human Kidney.. Sci Rep. 2016, 6: 33678. PMC 5030486
 . PMID 27649628. doi:10.1038/srep33678.
. PMID 27649628. doi:10.1038/srep33678.
- ^ Raizada MK, Ferreira AJ. ACE2: a new target for cardiovascular disease therapeutics.. J Cardiovasc Pharmacol. 2007, 50 (2): 112–9. PMID 17703127. doi:10.1097/FJC.0b013e3180986219.
- ^ Uhal BD, Li X, Xue A, Gao X, Abdul-Hafez A. Regulation of alveolar epithelial cell survival by the ACE-2/angiotensin 1-7/Mas axis.. Am J Physiol Lung Cell Mol Physiol. 2011, 301 (3): L269–74. PMC 3174737
 . PMID 21665960. doi:10.1152/ajplung.00222.2010.
. PMID 21665960. doi:10.1152/ajplung.00222.2010.
- ^ 17.0 17.1 Samavati L, Uhal BD. ACE2, Much More Than Just a Receptor for SARS-COV-2.. Front Cell Infect Microbiol. 2020, 10: 317. PMC 7294848
 . PMID 32582574. doi:10.3389/fcimb.2020.00317.
. PMID 32582574. doi:10.3389/fcimb.2020.00317.
- ^ Gopallawa I, Uhal BD. Angiotensin-(1-7)/mas inhibits apoptosis in alveolar epithelial cells through upregulation of MAP kinase phosphatase-2.. Am J Physiol Lung Cell Mol Physiol. 2016, 310 (3): L240–8. PMC 4888557
 . PMID 26637635. doi:10.1152/ajplung.00187.2015.
. PMID 26637635. doi:10.1152/ajplung.00187.2015.
- ^ Cabello-Verrugio C, Morales MG, Rivera JC, Cabrera D, Simon F. Renin-angiotensin system: an old player with novel functions in skeletal muscle.. Med Res Rev. 2015, 35 (3): 437–63. PMID 25764065. doi:10.1002/med.21343.
- ^ Winslow MA, Hall SE. Muscle wasting: A review of exercise, classical and non-classical RAS axes.. J Cell Mol Med. 2019, 23 (9): 5836–5845. PMC 6714228
 . PMID 31273946. doi:10.1111/jcmm.14412.
. PMID 31273946. doi:10.1111/jcmm.14412.
- ^ Morales MG, Abrigo J, Meneses C, Cisternas F, Simon F, Cabello-Verrugio C. Expression of the Mas receptor is upregulated in skeletal muscle wasting.. Histochem Cell Biol. 2015, 143 (2): 131–41. PMID 25208653. doi:10.1007/s00418-014-1275-1.
- ^ Riquelme C, Acuña MJ, Torrejón J, Rebolledo D, Cabrera D, Santos RA; et al. ACE2 is augmented in dystrophic skeletal muscle and plays a role in decreasing associated fibrosis.. PLoS One. 2014, 9 (4): e93449. PMC 3973684
 . PMID 24695436. doi:10.1371/journal.pone.0093449.
. PMID 24695436. doi:10.1371/journal.pone.0093449.
- ^ Schmoldt A, Benthe HF, Haberland G. Digitoxin metabolism by rat liver microsomes.. Biochem Pharmacol. 1975, 24 (17): 1639–41. doi:10.1371/journal.pone.0034747.
- ^ Lambert DW, Yarski M, Warner FJ, Thornhill P, Parkin ET, Smith AI, et al. Tumor necrosis factor-alpha convertase (ADAM17) mediates regulated ectodomain shedding of the severe-acute respiratory syndrome-coronavirus (SARS-CoV) receptor, angiotensin-converting enzyme-2 (ACE2). The Journal of Biological Chemistry. August 2005, 280 (34): 30113–9. PMID 15983030. doi:10.1074/jbc.M505111200.
- ^ Patel VB, Clarke N, Wang Z, Fan D, Parajuli N, Basu R, et al. Angiotensin II induced proteolytic cleavage of myocardial ACE2 is mediated by TACE/ADAM-17: a positive feedback mechanism in the RAS. Journal of Molecular and Cellular Cardiology. January 2014, 66: 167–76. PMID 24332999. doi:10.1016/j.yjmcc.2013.11.017.
- ^ Lambert DW, Clarke NE, Hooper NM, Turner AJ. Calmodulin interacts with angiotensin-converting enzyme-2 (ACE2) and inhibits shedding of its ectodomain.. FEBS Lett. 2008, 582 (2): 385–90. PMC 7094239
 . PMID 18070603. doi:10.1016/j.febslet.2007.11.085.
. PMID 18070603. doi:10.1016/j.febslet.2007.11.085.
- ^ Xu P, Derynck R. Direct activation of TACE-mediated ectodomain shedding by p38 MAP kinase regulates EGF receptor-dependent cell proliferation.. Mol Cell. 2010, 37 (4): 551–66. PMC 4240279
 . PMID 20188673. doi:10.1016/j.molcel.2010.01.034.
. PMID 20188673. doi:10.1016/j.molcel.2010.01.034.
- ^ Patel VB, Zhong JC, Grant MB, Oudit GY. Role of the ACE2/Angiotensin 1-7 Axis of the Renin-Angiotensin System in Heart Failure.. Circ Res. 2016, 118 (8): 1313–26. PMC 4939482
 . PMID 27081112. doi:10.1161/CIRCRESAHA.116.307708.
. PMID 27081112. doi:10.1161/CIRCRESAHA.116.307708.
- ^ Gene: ACE2, angiotensin I converting enzyme 2. National Center for Biotechnology Information (NCBI). U.S. National Library of Medicine. 2020-02-28.
- ^ Fehr AR, Perlman S. Coronaviruses: an overview of their replication and pathogenesis. Coronaviruses. Methods in Molecular Biology 1282. Springer New York. 2015: 1–23. ISBN 978-1-4939-2437-0. PMC 4369385
 . PMID 25720466. doi:10.1007/978-1-4939-2438-7_1.
. PMID 25720466. doi:10.1007/978-1-4939-2438-7_1. Many α-coronaviruses utilize aminopeptidase N (APN) as their receptor, SARS-CoV and HCoV-NL63 use angiotensin-converting enzyme 2 (ACE2) as their receptor, MHV enters through CEACAM1, and the recently identified MERS-CoV binds to dipeptidyl-peptidase 4 (DPP4) to gain entry into human cells (See Table 1 for a list of known CoV receptors).
- ^ Li F. Receptor recognition and cross-species infections of SARS coronavirus. Antiviral Research. October 2013, 100 (1): 246–54. PMC 3840050
 . PMID 23994189. doi:10.1016/j.antiviral.2013.08.014.
. PMID 23994189. doi:10.1016/j.antiviral.2013.08.014.
- ^ What are the official names of the disease and the virus that causes it?. Q&A on coronaviruses. World Health Organization. [22 February 2020]. (原始内容存档于5 March 2020).
- ^ Akhmerov Akbarshakh; Marban Eduardo. COVID-19 and the Heart. Circulation Research. 2020, 0 (10): 1443–1455. PMC 7188058
 . PMID 32252591. doi:10.1161/CIRCRESAHA.120.317055.
. PMID 32252591. doi:10.1161/CIRCRESAHA.120.317055.
- ^ Ou X, Liu Y, Lei X, Li P, Mi D, Ren L; et al. Characterization of spike glycoprotein of SARS-CoV-2 on virus entry and its immune cross-reactivity with SARS-CoV.. Nat Commun. 2020, 11 (1): 1620. PMC 7100515
 . PMID 32221306. doi:10.1038/s41467-020-15562-9.
. PMID 32221306. doi:10.1038/s41467-020-15562-9.
- ^ Wang H, Yang P, Liu K, Guo F, Zhang Y, Zhang G, Jiang C. SARS coronavirus entry into host cells through a novel clathrin- and caveolae-independent endocytic pathway. Cell Research. February 2008, 18 (2): 290–301. PMC 7091891
 . PMID 18227861. doi:10.1038/cr.2008.15.
. PMID 18227861. doi:10.1038/cr.2008.15.
- ^ Millet JK, Whittaker GR. Physiological and molecular triggers for SARS-CoV membrane fusion and entry into host cells. Virology. April 2018, 517: 3–8. PMC 7112017
 . PMID 29275820. doi:10.1016/j.virol.2017.12.015.
. PMID 29275820. doi:10.1016/j.virol.2017.12.015.
- ^ Huang IC, Bosch BJ, Li F, Li W, Lee KH, Ghiran S; et al. SARS coronavirus, but not human coronavirus NL63, utilizes cathepsin L to infect ACE2-expressing cells.. J Biol Chem. 2006, 281 (6): 3198–203. PMID 16339146. doi:10.1074/jbc.M508381200.
- ^ Andersen KG, Rambaut A, Lipkin WI, Holmes EC, Garry RF. The proximal origin of SARS-CoV-2.. Nat Med. 2020, 26 (4): 450–452. PMC 7095063
 . PMID 32284615. doi:10.1038/s41591-020-0820-9.
. PMID 32284615. doi:10.1038/s41591-020-0820-9.
参见
外部連結
- Human ACE2 genome location and ACE2 gene details page in the UCSC Genome Browser.
| |||||||
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||