T細胞
| 此條目需要編修,以確保標點符號使用恰當。 (2018年1月) |
| T細胞 | |
|---|---|
 人類T細胞的掃描電子顯微鏡圖 | |
 | |
| 基本資訊 | |
| 系統 | 免疫系統 |
| 標識字符 | |
| 拉丁文 | lymphocytus T |
| MeSH | D013601 |
| TH | H2.00.04.1.02007 |
| FMA | FMA:62870 |
| 《顯微解剖學術語》 [在維基數據上編輯] | |
T細胞(英語:T cell;T lymphocyte)是淋巴細胞的一種,在免疫反應中扮演著重要的角色,T是胸腺(thymus)的英文縮寫。T細胞在骨髓被製造出來之後,在胸腺內進行「新兵訓練」分化成熟為不同亞型的效應T細胞,成熟後就移居於周圍淋巴組織中開始工作。
T細胞膜表面分子有兩大類:T細胞受體(TCR)和分化群分子(CD molecules),其與T細胞的功能相關,也是T細胞的表面標誌(cell-surface marker),可以用以分離、鑑定不同亞群的T細胞[1]。
發育[編輯]
起源[編輯]
所有的T細胞都來源於造血幹細胞(HSC),造血幹細胞會分化為多能祖細胞(MPP),多能祖細胞又會分化為共同淋巴祖細胞(CLP),之後CLP只有三種分化路徑,即T細胞、B細胞和NK細胞[2]。 那些分化為T細胞的CLP將會隨著血流到達胸腺,並成為早期胸腺祖細胞(ETP),現在這些細胞既不表達CD4也不表達CD8[3]。這些細胞將經過一輪分裂之後會進入DN1階段。
TCR-β選擇[編輯]
在DN2階段(CD44+CD25+),細胞上調RAG1/2並重排TCR(T細胞受體)-β基因座,V-D-J序列和恆定區序列,目的是產生一個有功能的TCR-β鏈。當細胞經過DN3階段(CD44-CD25+)時,細胞將會和TCRβ一起表達一個未經重排的α-鏈(pre-Tα),如果重排後的β-鏈可以和pre-Tα形成二聚體,細胞將產生信號停止β-鏈的重排[4]。雖然這個信號需要pre-TCR在細胞膜上表達,不過它與pre-TCR和配體的結合無關。如果pre-TCR形成了,細胞會下調CD25並進入DN4階段(CD25-CD44-),這些細胞將繼續分裂並重排TCRα的基因座。
陽性選擇[編輯]
雙陽性(CD4+/CD8+)的T細胞會向胸腺皮層深處遷移,並會接觸到胸腺皮層上皮細胞表面的「自體抗原」(self-antigens)。這些自體抗原結合在胸腺上皮細胞表面的MHC分子上,只有與胸腺細胞的MHC分子表現出足夠強的結合力的T細胞,才能接收到必要的「存活信號」,而無法接收到足夠「存活信號」的T細胞將會凋亡。在這個持續幾天的陽性選擇過程中,大部分的T細胞都會死去[5]。
一個T細胞的命運就在陽性選擇的過程中被決定。在雙陽性(CD4+/CD8+)T細胞中,能夠與MHC Ⅱ類分子結合得較好的將成為CD4+細胞,而和MHC Ⅰ類分子有更高親和力的將成為CD8+細胞。將成為CD4+細胞的細胞將會逐漸下調自己的CD8,最終成為單陽性的CD4+細胞[6]。
陰性選擇[編輯]
在陽性選擇中存活下來的T細胞將會向胸腺皮質邊緣和髓質區遷移;在髓質區,它們又會接觸到胸腺髓質上皮細胞(mTECs)表面的自體抗原[7]。mTECs會在它們的MHC Ⅰ類分子上呈遞來自全身各個組織的自體抗原。一些mTECs被胸腺樹狀細胞吞噬,它們的自體抗原就會呈遞在樹狀細胞的MHC Ⅱ類分子上(經過了陽性選擇的CD4+細胞只能識別MHC Ⅱ類分子)。在這裡,與自體抗原表現出過強的親和力的T細胞會接收到凋亡信號並凋亡(在這些細胞中也有一部分會成為調節T細胞),存活下來的細胞就作為成熟的初級T細胞離開胸腺[8]。這一過程是中樞免疫耐受的重要組成部分,其意義在於篩選掉可能對自體抗原產生反應的T細胞,從而避免自體免疫疾病的發生。
胸腺輸出[編輯]
經過陽性選擇和陰性選擇,最初到達胸腺的T細胞中有98%死亡,存活下來的2%成為了具有成熟免疫功能的T細胞。胸腺產生成熟T細胞的數量大致隨著個體衰老而減少,在中年人的體內,胸腺的大小平均每年縮小3%[9]。所以,對中老年人而言,外周T細胞的增殖和再生對於免疫系統的意義更大。
分類[編輯]
T細胞根據功能的差異被分為幾個亞型。雖然在胸腺中就分化出了CD4+和CD8+兩者,但是在外周T細胞還會發生進一步的分化。
常規適應性T細胞[編輯]
輔助性CD4+ T細胞[編輯]
輔助性T細胞(TH細胞)對其他淋巴細胞的活動起輔助作用,包括B細胞向漿細胞和記憶B細胞的發育,以及細胞毒性T細胞和巨噬細胞的激活。它們也被稱為CD4+ T細胞,因為它們的細胞表面有CD4蛋白的表達。輔助T細胞在遇到抗原呈遞細胞(APC)表面MHC-II分子結合的外部抗原時被激活,一旦被激活就會快速分裂並開始分泌調節免疫反應的細胞因子。輔助T細胞在受到不同細胞因子刺激的情況下,也會進一步分化成不同亞型的輔助T細胞[10]。它們是已知的HIV病毒的目標細胞,在愛滋病發病時會急劇減少。濾泡輔助性T細胞是參與體液應答的CD4陽性輔助性T細胞的一個亞群[11],屬一類專門為B細胞提供幫助的CD4+ T細胞[12]。
調節性CD4+ T細胞[編輯]
調節性T細胞(Treg細胞)舊稱抑制性T細胞,對於免疫耐受至關重要。它們的主要工作就是及時有效的結束免疫反應,以及抑制那些從陰性篩選中逃逸的自體免疫T細胞,防止免疫反應對抑制性機體自身造成過度損害。
調節性T細胞既可以在胸腺中發育分化完成,稱為胸腺調節T細胞;也可以在外周組織受免疫反應誘導分化,稱為外周調節T細胞[13]。兩者都表達FOXP3作為其細胞表面標誌物,FOXP3基因的突變會影響調節T細胞的發育,並誘發致命的自體免疫病IPEX。
其他幾類不表達FOXP3基因的T細胞具有免疫抑制作用,例如Tr1細胞和Th3細胞。Tr1與IL-10相關,Th3與TGF-beta相關。最近,Th17細胞也被列入此類免疫抑制細胞之中[14]。
細胞毒性CD8+ T細胞[編輯]
細胞毒性T細胞(CTLs, killer T cells)負責殺傷被病毒感染的細胞和癌細胞,在對器官移植的免疫排斥中也有參與。其特點在於細胞表面的CD8蛋白質。它通過識別所有有核細胞表面的MHC-I分子上的短肽抗原,來分辨正常細胞和應殺傷的異常細胞。細胞毒性T細胞還可分泌重要的細胞因子IL-2和IFNγ,來影響其他免疫細胞的功能,特別是巨噬細胞和NK細胞。
記憶性T細胞[編輯]
還未結合過外部抗原的初級T細胞,一旦結合了抗原呈遞細胞表面MHC分子所包裹的外部抗原,就會開始增殖分化為效應T細胞和記憶T細胞(其他信號適當的共刺激對這一過程也是必要的)。曾經,人們認為記憶T細胞只分為中央記憶T細胞和效應記憶T細胞[15]。但是之後,新的記憶T細胞種類不斷被發現,例如組織駐留記憶T細胞 (Trm)等等。記憶T細胞的共同特點在於其壽命較長(可長達數十年),而且在識別到特定抗原時可以快速分裂為大量的效應T細胞。通過這樣的方式,記憶T細胞就為人體的免疫系統保存了對之前感染過病原體的「記憶」。記憶T既可以是CD4+也可以是CD8+,一般會表達CD45RO[16]。
類固有T細胞[編輯]
自然殺傷T細胞[編輯]
自然殺傷T細胞(NKT細胞),請不要把它和固有免疫中的自然殺傷細胞(NK細胞)混淆。 與一般T細胞識別MHC分子上的肽鏈抗原不同,NKT識別的是CD1d分子上結合的糖蛋白抗原。被激活後,它們可以執行類似輔助T細胞和細胞毒性T細胞的功能,即釋放細胞因子和細胞毒素。有證據表明,它們能夠識別並殺滅某些腫瘤細胞和被疱疹病毒感染的細胞[17]。
黏膜相關的不變T細胞[編輯]
黏膜相關的不變T細胞(MAIT)具有固有免疫效應細胞的特質[18][19]。在人體內,MAIT細胞分布於血液、肝臟、肺部、黏膜,具有抵禦微生物感染的能力[18]。MHC-I的類似物,MR1,可以向MAIT細胞呈遞細菌產生的代謝物抗原[20][21][22]。接受了MR1呈遞的外部抗原後,MAIT細胞可以釋放促炎症細胞因子並裂解被細菌感染的細胞[18][22]。MAIT也可以通過不依靠MR1的信號通路激活[22]。除了表現出類似固有免疫的功能外,MAIT細胞也輔助獲得性免疫反應,甚至表現出類似記憶細胞的特徵[18]。此外,MAIT也被認為在自體免疫病中發揮作用,如多發性硬化、風濕和炎症性腸病[23][24],雖然決定性的證據還有待發現[25][26][27][28]。
γδ T細胞[編輯]
γδ T細胞代表了T細胞中一小部分不表達αβ-TCR而表達γδ-TCR的類型,在小鼠和人體內僅占全部T細胞的2%;在兔子、綿羊和雞體內,γδ T細胞占全部T細胞的比例則可能高達60%。它們主要分布在腸道黏膜,作為一類上皮內淋巴細胞。關於其抗原識別的細節我們仍知之甚少,不過似乎γδ T細胞並不受限於MHC分子的呈遞。特別的是,γδ T細胞能夠對一類磷酸類抗原做出快速的反應,而這類抗原物質在各種細胞(細菌、植物、癌細胞等)中都有發現。
活化[編輯]
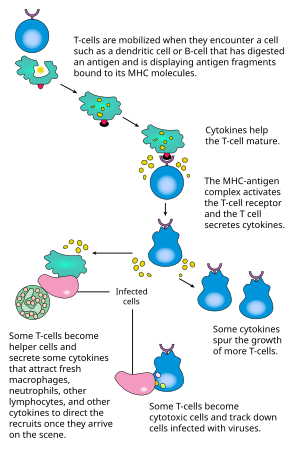
CD4+ T細胞的激活需要T細胞上的TCR和共受體(CD28或ICOS),抗原呈遞細胞上的MHCII和共激活分子兩對分子的分別,同時結合。僅其中一對的結合,無法產生有效的T細胞激活。理想的CD8+ T細胞激活則依賴於CD4+ T細胞的信號轉導[30]。CD4+細胞可以在初級CD8 T細胞的初次免疫應答中給予幫助,並且在急性感染的後期維持CD8+ 記憶T細胞的活性。所以,CD4+ T的激活對於CD8+ T細胞的活動是有利的[31][32][33]。
相比於MHC分子上的抗原,抗原呈遞細胞的共激活分子一般是由病原體的副產物、熱休克蛋白或者壞死的細胞碎片誘導表達的。共刺激機制被認為可以避免自體免疫的發生,因為即使T細胞錯誤地結合了自體抗原,也可能因為沒有受到合適的共刺激而無法正常活化。一旦T細胞被正確地活化,它的細胞表面蛋白表達就會發生巨大的改變,活化T細胞的標誌蛋白包括CD69,CD71,CD25 (也是調節T細胞的標誌)和HLA-DR (人類T細胞的特異標誌)。CTLA-4在活化T細胞表面的上調,對共激活受體有競爭性抑制作用,可以避免活化T細胞的過度活化。活化T細胞的表面糖基化情況也有改變[34]。
T細胞受體(TCR)是由幾種蛋白質組合成的複合體。TCR的兩個主要組分是由兩個獨立基因分別編碼的TCRα和TCRβ,其他的組分包括CD3家族的蛋白:CD3εγ和CD3εδ的異二聚體,以及最重要的CD3ζ同二聚體。CD3ζ同二聚體上共有6個ITAM基序,可被磷酸化並啟動一系列級聯反應,導致TCR複合體的聚集。
雖然在絕大部分情況下T細胞活化都依賴於TCR對抗原的識別,其他的活化途徑也有被發現,例如細胞毒性T細胞可以被其他CD8 T細胞識別並導致自身的極化[35]。T細胞活化的過程也受到活性氧類物質的影響[36]。
抗原識別[編輯]
T細胞的主要特點就是能夠分辨正常細胞和異常細胞的能力[37]。不論是正常細胞還是異常細胞,都會表達大量的MHC-抗原多肽複合體(pMHC)。雖然T細胞與正常細胞的pMHC有一定結合力,但是T細胞並不會被激活;但即使異常細胞的pMHC與正常細胞只有細微的差別,也能夠刺激T細胞發生免疫反應。這樣對不同抗原完全不同的反應特徵稱為T細胞的抗原識別,關於這一機理實現的具體細節如今仍然沒有定論[38]。
臨床意義[編輯]
缺陷[編輯]
T細胞缺陷可能意味著T細胞數量的減少或者T細胞功能的缺失。完全的T細胞缺陷可能來自於一些遺傳因素,例如嚴重複合型免疫缺乏症(SCID)、歐門氏症候群或軟骨毛髮發育不全。[46]部分的T細胞缺陷可能是由於獲得性免疫缺陷症候群(AIDS)、遺傳性的迪喬治症候群(DGS)、染色體斷裂症候群(CBSs),或者B細胞和T細胞的複合缺陷,例如毛細血管擴張性運動失調 (AT) 和歐德里症候群(Wiskott–Aldrich syndrome)[39]。
T細胞缺陷患者面臨的主要風險,主要是一些細胞內病原體,例如單純疱疹病毒、分枝桿菌和李斯特菌。同時,真菌感染在T細胞缺陷患者身上往往也很常見且嚴重[40]。
癌症[編輯]
T細胞癌變誘發的腫瘤稱為T細胞淋巴瘤,在非霍奇金淋巴瘤中約占10%[41]。
耗竭[編輯]
T細胞耗竭是一種T細胞功能失常的狀態,其表現為進展性的功能喪失、基因表達譜的變化、和抑制性細胞因子的持續分泌。T細胞耗竭可能發生於慢性感染、敗血症、癌症的進程中[42]。耗竭的T細胞即使再次暴露於抗原刺激之中也無法恢復正常功能[43]。
慢性感染和敗血症[編輯]
T細胞耗竭的直接原因包括持續的抗原刺激、以及CD4細胞的缺失[44]。長時間的抗原暴露和高病毒負載可以加重T細胞耗竭的程度。2-4周的持續抗原暴露就可導致T細胞耗竭[45]。另一個可以導致T細胞耗竭的因素是包括PD-1在內的一系列抑制性受體[46][47]。細胞因子IL-10或TGF-β也可以導致耗竭[48][49]。調節T細胞因為可以分泌IL-10和TGF-β,也與T細胞耗竭相關[50]。在阻斷PD-1受體並減少調節T細胞數量後,T細胞耗竭的情況可以得到反轉[51]。[58]在敗血症中,抑制性的細胞因子風暴也會造成T細胞耗竭[52][53]。現在已有致力於通過阻斷抑制性受體的方式來治療敗血症的療法研究[54][55][56]。
器官移植[編輯]
與感染時的情況類似,器官移植帶來的持續異種抗原暴露也會造成T細胞耗竭[57]。腎移植後,T細胞應答能力會隨時間減弱[58]。這些數據說明T細胞耗竭導致的CD8+ T細胞數量減少可能是器官移植耐受中的重要一環[59]。已有幾項研究證明了慢性感染對器官移植後的免疫耐受和長期生存有利,而T細胞耗竭起著一定介導的作用[60][61][62]。雖然已有T細胞耗竭對器官移植有利的證據,但是T細胞耗竭同時帶來的感染和癌變風險依然不能忽視[63]。
癌症[編輯]
在癌症進程中,T細胞耗竭顯然對癌組織的存活有利。已有研究證明癌細胞和一些癌症相關細胞可以主動地誘導T細胞耗竭的發生[64][65] [66]。在白血病中,T細胞耗竭也與其復發相關[67]。一些研究甚至提出可以基於T細胞抑制性受體PD-1的表達狀態來預測白血病復發的情況[68]。由於免疫抑制性受體與T細胞耗竭以及癌症之間的關係,近年來有大量的研究和臨床試驗致力於通過阻斷免疫抑制性受體來治療癌症,其中有一些已經被認定有效並投入臨床使用[69][70]。
參見[編輯]
參考文獻[編輯]
- ^ Janeway, Charles. Immunobiology: the immune system in health and disease 5th. New York: Garland Pub. 2001 [2020-02-12]. ISBN 978-0-8153-3642-6. OCLC 45708106. (原始內容存檔於2019-10-17).
- ^ Kondo, Motonari. One Niche to Rule Both Maintenance and Loss of Stemness in HSCs. Immunity. 2016-12-20, 45 (6): 1177–1179 [2020-02-23]. ISSN 1097-4180. PMID 28002722. doi:10.1016/j.immuni.2016.12.003. (原始內容存檔於2020-03-18).
- ^ Osborne, Lisa C.; Dhanji, Salim; Snow, Jonathan W.; Priatel, John J.; Ma, Melissa C.; Miners, M. Jill; Teh, Hung-Sia; Goldsmith, Mark A.; Abraham, Ninan. Impaired CD8 T cell memory and CD4 T cell primary responses in IL-7R alpha mutant mice. The Journal of Experimental Medicine. 2007-03-19, 204 (3): 619–631 [2020-02-23]. ISSN 0022-1007. PMC 2137912
 . PMID 17325202. doi:10.1084/jem.20061871. (原始內容存檔於2020-03-18).
. PMID 17325202. doi:10.1084/jem.20061871. (原始內容存檔於2020-03-18).
- ^ Murphy, Kenneth (Kenneth M.); Walport, Mark.; Janeway, Charles. Janeway's immunobiology 8th. New York: Garland Science. 2012: 301–305. ISBN 978-0-8153-4243-4. OCLC 733935898.
- ^ Starr, Timothy K.; Jameson, Stephen C.; Hogquist, Kristin A. Positive and negative selection of T cells. Annual Review of Immunology. 2003, 21: 139–176 [2020-02-23]. ISSN 0732-0582. PMID 12414722. doi:10.1146/annurev.immunol.21.120601.141107. (原始內容存檔於2020-03-18).
- ^ Zerrahn, J.; Held, W.; Raulet, D. H. The MHC reactivity of the T cell repertoire prior to positive and negative selection. Cell. 1997-03-07, 88 (5): 627–636 [2020-02-23]. ISSN 0092-8674. PMID 9054502. doi:10.1016/s0092-8674(00)81905-4. (原始內容存檔於2020-03-18).
- ^ Hinterberger, Maria; Aichinger, Martin; Prazeres da Costa, Olivia; Voehringer, David; Hoffmann, Reinhard; Klein, Ludger. Autonomous role of medullary thymic epithelial cells in central CD4(+) T cell tolerance. Nature Immunology. 2010-06, 11 (6): 512–519 [2020-02-23]. ISSN 1529-2916. PMID 20431619. doi:10.1038/ni.1874. (原始內容存檔於2020-03-18).
- ^ Pekalski, Marcin L.; García, Arcadio Rubio; Ferreira, Ricardo C.; Rainbow, Daniel B.; Smyth, Deborah J.; Mashar, Meghavi; Brady, Jane; Savinykh, Natalia; Dopico, Xaquin Castro. Neonatal and adult recent thymic emigrants produce IL-8 and express complement receptors CR1 and CR2. JCI insight. 2017-08-17, 2 (16) [2020-02-23]. ISSN 2379-3708. PMC 5621870
 . PMID 28814669. doi:10.1172/jci.insight.93739. (原始內容存檔於2020-03-18).
. PMID 28814669. doi:10.1172/jci.insight.93739. (原始內容存檔於2020-03-18).
- ^ Haynes, B. F.; Markert, M. L.; Sempowski, G. D.; Patel, D. D.; Hale, L. P. The role of the thymus in immune reconstitution in aging, bone marrow transplantation, and HIV-1 infection. Annual Review of Immunology. 2000, 18: 529–560 [2020-02-23]. ISSN 0732-0582. PMID 10837068. doi:10.1146/annurev.immunol.18.1.529. (原始內容存檔於2020-03-18).
- ^ Gutcher, Ilona; Becher, Burkhard. APC-derived cytokines and T cell polarization in autoimmune inflammation. The Journal of Clinical Investigation. 2007-05, 117 (5): 1119–1127 [2020-02-23]. ISSN 0021-9738. PMC 1857272
 . PMID 17476341. doi:10.1172/JCI31720. (原始內容存檔於2020-03-18).
. PMID 17476341. doi:10.1172/JCI31720. (原始內容存檔於2020-03-18).
- ^ Crotty S (2014) T follicular helper cell differentiation, function, and roles in disease.Immunity 41:529–542.
- ^ 何嵐; 孫兵. 滤泡辅助性T细胞分化和功能的研究进展 (PDF). 生命科學. 2016, 28 (2): 146 [2023-05-05]. doi:10.13376/j.cbls/2016021. (原始內容存檔 (PDF)於2023-05-05).
- ^ Abbas, Abul K.; Benoist, Christophe; Bluestone, Jeffrey A.; Campbell, Daniel J.; Ghosh, Sankar; Hori, Shohei; Jiang, Shuiping; Kuchroo, Vijay K.; Mathis, Diane. Regulatory T cells: recommendations to simplify the nomenclature. Nature Immunology. 2013-04, 14 (4): 307–308 [2020-02-23]. ISSN 1529-2916. PMID 23507634. doi:10.1038/ni.2554. (原始內容存檔於2020-03-18).
- ^ Singh, Bhagirath; Schwartz, Jordan Ari; Sandrock, Christian; Bellemore, Stacey M.; Nikoopour, Enayat. Modulation of autoimmune diseases by interleukin (IL)-17 producing regulatory T helper (Th17) cells. The Indian Journal of Medical Research. 2013-11, 138 (5): 591–594 [2020-02-23]. ISSN 0971-5916. PMC 3928692
 . PMID 24434314. (原始內容存檔於2020-03-18).
. PMID 24434314. (原始內容存檔於2020-03-18).
- ^ Sallusto, F.; Lenig, D.; Förster, R.; Lipp, M.; Lanzavecchia, A. Two subsets of memory T lymphocytes with distinct homing potentials and effector functions. Nature. 1999-10-14, 401 (6754): 708–712 [2020-02-23]. ISSN 0028-0836. PMID 10537110. doi:10.1038/44385. (原始內容存檔於2020-03-18).
- ^ Akbar, A. N.; Terry, L.; Timms, A.; Beverley, P. C.; Janossy, G. Loss of CD45R and gain of UCHL1 reactivity is a feature of primed T cells. Journal of Immunology (Baltimore, Md.: 1950). 1988-04-01, 140 (7): 2171–2178 [2020-02-23]. ISSN 0022-1767. PMID 2965180. (原始內容存檔於2020-03-18).
- ^ Mallevaey, Thierry; Fontaine, Josette; Breuilh, Laetitia; Paget, Christophe; Castro-Keller, Alexandre; Vendeville, Catherine; Capron, Monique; Leite-de-Moraes, Maria; Trottein, François. Invariant and noninvariant natural killer T cells exert opposite regulatory functions on the immune response during murine schistosomiasis. Infection and Immunity. 2007-05, 75 (5): 2171–2180 [2020-02-23]. ISSN 0019-9567. PMC 1865739
 . PMID 17353286. doi:10.1128/IAI.01178-06. (原始內容存檔於2020-03-18).
. PMID 17353286. doi:10.1128/IAI.01178-06. (原始內容存檔於2020-03-18).
- ^ 18.0 18.1 18.2 18.3 Napier RJ, Adams EJ, Gold MC, Lewinsohn DM. The Role of Mucosal Associated Invariant T Cells in Antimicrobial Immunity. Frontiers in Immunology. 2015-07-06, 6: 344. PMC 4492155
 . PMID 26217338. doi:10.3389/fimmu.2015.00344.
. PMID 26217338. doi:10.3389/fimmu.2015.00344.
- ^ Gold MC, Lewinsohn DM. Mucosal associated invariant T cells and the immune response to infection. Microbes and Infection. August 2011, 13 (8–9): 742–8. PMC 3130845
 . PMID 21458588. doi:10.1016/j.micinf.2011.03.007.
. PMID 21458588. doi:10.1016/j.micinf.2011.03.007.
- ^ Eckle SB, Corbett AJ, Keller AN, Chen Z, Godfrey DI, Liu L, Mak JY, Fairlie DP, Rossjohn J, McCluskey J. Recognition of Vitamin B Precursors and Byproducts by Mucosal Associated Invariant T Cells. The Journal of Biological Chemistry. December 2015, 290 (51): 30204–11. PMC 4683245
 . PMID 26468291. doi:10.1074/jbc.R115.685990.
. PMID 26468291. doi:10.1074/jbc.R115.685990.
- ^ Ussher JE, Klenerman P, Willberg CB. Mucosal-associated invariant T-cells: new players in anti-bacterial immunity. Frontiers in Immunology. 2014-10-08, 5: 450. PMC 4189401
 . PMID 25339949. doi:10.3389/fimmu.2014.00450.
. PMID 25339949. doi:10.3389/fimmu.2014.00450.
- ^ 22.0 22.1 22.2 Howson LJ, Salio M, Cerundolo V. MR1-Restricted Mucosal-Associated Invariant T Cells and Their Activation during Infectious Diseases. Frontiers in Immunology. 2015-06-16, 6: 303. PMC 4468870
 . PMID 26136743. doi:10.3389/fimmu.2015.00303.
. PMID 26136743. doi:10.3389/fimmu.2015.00303.
- ^ Hinks TS. Mucosal-associated invariant T cells in autoimmunity, immune-mediated diseases and airways disease. Immunology. May 2016, 148 (1): 1–12. PMC 4819138
 . PMID 26778581. doi:10.1111/imm.12582.
. PMID 26778581. doi:10.1111/imm.12582.
- ^ Bianchini E, De Biasi S, Simone AM, Ferraro D, Sola P, Cossarizza A, Pinti M. Invariant natural killer T cells and mucosal-associated invariant T cells in multiple sclerosis. Immunology Letters. March 2017, 183: 1–7. PMID 28119072. doi:10.1016/j.imlet.2017.01.009.
- ^ Serriari NE, Eoche M, Lamotte L, Lion J, Fumery M, Marcelo P, Chatelain D, Barre A, Nguyen-Khac E, Lantz O, Dupas JL, Treiner E. Innate mucosal-associated invariant T (MAIT) cells are activated in inflammatory bowel diseases. Clinical and Experimental Immunology. May 2014, 176 (2): 266–74. PMC 3992039
 . PMID 24450998. doi:10.1111/cei.12277.
. PMID 24450998. doi:10.1111/cei.12277.
- ^ Huang S, Martin E, Kim S, Yu L, Soudais C, Fremont DH, Lantz O, Hansen TH. MR1 antigen presentation to mucosal-associated invariant T cells was highly conserved in evolution. Proceedings of the National Academy of Sciences of the United States of America. May 2009, 106 (20): 8290–5. Bibcode:2009PNAS..106.8290H. PMC 2688861
 . PMID 19416870. doi:10.1073/pnas.0903196106.
. PMID 19416870. doi:10.1073/pnas.0903196106.
- ^ Chua WJ, Hansen TH. Bacteria, mucosal-associated invariant T cells and MR1. Immunology and Cell Biology. November 2010, 88 (8): 767–9. PMID 20733595. doi:10.1038/icb.2010.104.
- ^ Kjer-Nielsen L, Patel O, Corbett AJ, Le Nours J, Meehan B, Liu L, Bhati M, Chen Z, Kostenko L, Reantragoon R, Williamson NA, Purcell AW, Dudek NL, McConville MJ, O'Hair RA, Khairallah GN, Godfrey DI, Fairlie DP, Rossjohn J, McCluskey J. MR1 presents microbial vitamin B metabolites to MAIT cells (PDF). Nature. November 2012, 491 (7426): 717–23. Bibcode:2012Natur.491..717K. PMID 23051753. doi:10.1038/nature11605.
- ^ The NIAID resource booklet "Understanding the Immune System (pdf)" (頁面存檔備份,存於網際網路檔案館).
- ^ Williams, Matthew A.; Bevan, Michael J. Effector and memory CTL differentiation. Annual Review of Immunology. 2007, 25: 171–192 [2020-02-23]. ISSN 0732-0582. PMID 17129182. doi:10.1146/annurev.immunol.25.022106.141548. (原始內容存檔於2020-03-18).
- ^ Janssen EM, Lemmens EE, Wolfe T, Christen U, von Herrath MG, Schoenberger SP. CD4+ T cells are required for secondary expansion and memory in CD8+ T lymphocytes. Nature. February 2003, 421 (6925): 852–6. Bibcode:2003Natur.421..852J. PMID 12594515. doi:10.1038/nature01441.
- ^ Shedlock DJ, Shen H. Requirement for CD4 T cell help in generating functional CD8 T cell memory. Science. April 2003, 300 (5617): 337–9. Bibcode:2003Sci...300..337S. PMID 12690201. doi:10.1126/science.1082305.
- ^ Sun JC, Williams MA, Bevan MJ. CD4+ T cells are required for the maintenance, not programming, of memory CD8+ T cells after acute infection. Nature Immunology. September 2004, 5 (9): 927–33. PMC 2776074
 . PMID 15300249. doi:10.1038/ni1105.
. PMID 15300249. doi:10.1038/ni1105.
- ^ Maverakis E, Kim K, Shimoda M, Gershwin M, Patel F, Wilken R, Raychaudhuri S, Ruhaak LR, Lebrilla CB. Glycans in the immune system and The Altered Glycan Theory of Autoimmunity. J Autoimmun. 2015, 57 (6): 1–13. PMC 4340844
 . PMID 25578468. doi:10.1016/j.jaut.2014.12.002.
. PMID 25578468. doi:10.1016/j.jaut.2014.12.002.
- ^ Milstein O, Hagin D, Lask A, Reich-Zeliger S, Shezen E, Ophir E, Eidelstein Y, Afik R, Antebi YE, Dustin ML, Reisner Y. CTLs respond with activation and granule secretion when serving as targets for T cell recognition. Blood. January 2011, 117 (3): 1042–52. PMC 3035066
 . PMID 21045195. doi:10.1182/blood-2010-05-283770.
. PMID 21045195. doi:10.1182/blood-2010-05-283770.
- ^ Belikov AV, Schraven B, Simeoni L. T cells and reactive oxygen species. Journal of Biomedical Science. October 2015, 22: 85. PMC 4608155
 . PMID 26471060. doi:10.1186/s12929-015-0194-3.
. PMID 26471060. doi:10.1186/s12929-015-0194-3.
- ^ Feinerman O, Germain RN, Altan-Bonnet G. Quantitative challenges in understanding ligand discrimination by alphabeta T cells. Mol. Immunol. 2008, 45 (3): 619–31. PMC 2131735
 . PMID 17825415. doi:10.1016/j.molimm.2007.03.028.
. PMID 17825415. doi:10.1016/j.molimm.2007.03.028.
- ^ Dushek O, van der Merwe PA. An induced rebinding model of antigen discrimination. Trends Immunol. 2014, 35 (4): 153–8. PMC 3989030
 . PMID 24636916. doi:10.1016/j.it.2014.02.002.
. PMID 24636916. doi:10.1016/j.it.2014.02.002.
- ^ Medscape > T-cell Disorders (頁面存檔備份,存於網際網路檔案館). Author: Robert A Schwartz, MD, MPH; Chief Editor: Harumi Jyonouchi, MD. Updated: May 16, 2011
- ^ Bannister, Barbara A.; Jones, Jane. Infection : microbiology and management 3rd. Malden, Mass.: Blackwell Pub. 2006: 435. ISBN 978-1-4443-2393-1. OCLC 592756309.
- ^ The Lymphomas (PDF). The Leukemia & Lymphoma Society: 2. May 2006 [2008-04-07]. (原始內容 (PDF)存檔於2008-07-06).
- ^ Yi, John S.; Cox, Maureen A.; Zajac, Allan J. T-cell exhaustion: characteristics, causes and conversion. Immunology. 2010-04, 129 (4): 474–481 [2020-02-23]. ISSN 1365-2567. PMC 2842494
 . PMID 20201977. doi:10.1111/j.1365-2567.2010.03255.x. (原始內容存檔於2020-03-26).
. PMID 20201977. doi:10.1111/j.1365-2567.2010.03255.x. (原始內容存檔於2020-03-26).
- ^ Wang, Qin; Pan, Wen; Liu, Yanan; Luo, Jinzhuo; Zhu, Dan; Lu, Yinping; Feng, Xuemei; Yang, Xuecheng; Dittmer, Ulf. Hepatitis B Virus-Specific CD8+ T Cells Maintain Functional Exhaustion after Antigen Reexposure in an Acute Activation Immune Environment. Frontiers in Immunology. 2018, 9: 219 [2020-02-23]. ISSN 1664-3224. PMC 5816053
 . PMID 29483916. doi:10.3389/fimmu.2018.00219. (原始內容存檔於2020-03-26).
. PMID 29483916. doi:10.3389/fimmu.2018.00219. (原始內容存檔於2020-03-26).
- ^ Matloubian, M.; Concepcion, R. J.; Ahmed, R. CD4+ T cells are required to sustain CD8+ cytotoxic T-cell responses during chronic viral infection. Journal of Virology. 1994-12, 68 (12): 8056–8063 [2020-02-23]. ISSN 0022-538X. PMC 237269
 . PMID 7966595. (原始內容存檔於2020-03-18).
. PMID 7966595. (原始內容存檔於2020-03-18).
- ^ Angelosanto, Jill M.; Blackburn, Shawn D.; Crawford, Alison; Wherry, E. John. Progressive loss of memory T cell potential and commitment to exhaustion during chronic viral infection. Journal of Virology. 2012-08, 86 (15): 8161–8170 [2020-02-23]. ISSN 1098-5514. PMC 3421680
 . PMID 22623779. doi:10.1128/JVI.00889-12. (原始內容存檔於2020-03-18).
. PMID 22623779. doi:10.1128/JVI.00889-12. (原始內容存檔於2020-03-18).
- ^ Wherry EJ. T cell exhaustion. Nature Immunology. June 2011, 12 (6): 492–9. PMID 21739672. doi:10.1038/ni.2035.
- ^ Okagawa T, Konnai S, Nishimori A, Maekawa N, Goto S, Ikebuchi R, Kohara J, Suzuki Y, Yamada S, Kato Y, Murata S, Ohashi K. + T cells during bovine leukemia virus infection. Veterinary Research. June 2018, 49 (1): 50. PMC 6006750
 . PMID 29914540. doi:10.1186/s13567-018-0543-9 (英語).
. PMID 29914540. doi:10.1186/s13567-018-0543-9 (英語).
- ^ Brooks DG, Trifilo MJ, Edelmann KH, Teyton L, McGavern DB, Oldstone MB. Interleukin-10 determines viral clearance or persistence in vivo. Nature Medicine. November 2006, 12 (11): 1301–9. PMC 2535582
 . PMID 17041596. doi:10.1038/nm1492.
. PMID 17041596. doi:10.1038/nm1492.
- ^ Tinoco R, Alcalde V, Yang Y, Sauer K, Zuniga EI. Cell-intrinsic transforming growth factor-beta signaling mediates virus-specific CD8+ T cell deletion and viral persistence in vivo. Immunity. July 2009, 31 (1): 145–57. PMC 3039716
 . PMID 19604493. doi:10.1016/j.immuni.2009.06.015.
. PMID 19604493. doi:10.1016/j.immuni.2009.06.015.
- ^ Veiga-Parga T, Sehrawat S, Rouse BT. Role of regulatory T cells during virus infection. Immunological Reviews. September 2013, 255 (1): 182–96. PMC 3748387
 . PMID 23947355. doi:10.1111/imr.12085.
. PMID 23947355. doi:10.1111/imr.12085.
- ^ Penaloza-MacMaster P, Kamphorst AO, Wieland A, Araki K, Iyer SS, West EE, O'Mara L, Yang S, Konieczny BT, Sharpe AH, Freeman GJ, Rudensky AY, Ahmed R. Interplay between regulatory T cells and PD-1 in modulating T cell exhaustion and viral control during chronic LCMV infection. The Journal of Experimental Medicine. August 2014, 211 (9): 1905–18. PMC 4144726
 . PMID 25113973. doi:10.1084/jem.20132577.
. PMID 25113973. doi:10.1084/jem.20132577.
- ^ Otto GP, Sossdorf M, Claus RA, Rödel J, Menge K, Reinhart K, Bauer M, Riedemann NC. The late phase of sepsis is characterized by an increased microbiological burden and death rate. Critical Care. July 2011, 15 (4): R183. PMC 3387626
 . PMID 21798063. doi:10.1186/cc10332 (英語).
. PMID 21798063. doi:10.1186/cc10332 (英語).
- ^ Boomer JS, To K, Chang KC, Takasu O, Osborne DF, Walton AH, Bricker TL, Jarman SD, Kreisel D, Krupnick AS, Srivastava A, Swanson PE, Green JM, Hotchkiss RS. Immunosuppression in patients who die of sepsis and multiple organ failure. JAMA. December 2011, 306 (23): 2594–605. PMC 3361243
 . PMID 22187279. doi:10.1001/jama.2011.1829.
. PMID 22187279. doi:10.1001/jama.2011.1829.
- ^ Shindo Y, McDonough JS, Chang KC, Ramachandra M, Sasikumar PG, Hotchkiss RS. Anti-PD-L1 peptide improves survival in sepsis. The Journal of Surgical Research. February 2017, 208: 33–39. PMC 5535083
 . PMID 27993215. doi:10.1016/j.jss.2016.08.099.
. PMID 27993215. doi:10.1016/j.jss.2016.08.099.
- ^ Patera AC, Drewry AM, Chang K, Beiter ER, Osborne D, Hotchkiss RS. Frontline Science: Defects in immune function in patients with sepsis are associated with PD-1 or PD-L1 expression and can be restored by antibodies targeting PD-1 or PD-L1. Journal of Leukocyte Biology. December 2016, 100 (6): 1239–1254. PMC 5110001
 . PMID 27671246. doi:10.1189/jlb.4hi0616-255r.
. PMID 27671246. doi:10.1189/jlb.4hi0616-255r.
- ^ Wei Z, Li P, Yao Y, Deng H, Yi S, Zhang C, Wu H, Xie X, Xia M, He R, Yang XP, Tang ZH. Alpha-lactose reverses liver injury via blockade of Tim-3-mediated CD8 apoptosis in sepsis. Clinical Immunology. July 2018, 192: 78–84. PMID 29689313. doi:10.1016/j.clim.2018.04.010.
- ^ Wells AD, Li XC, Strom TB, Turka LA. The role of peripheral T-cell deletion in transplantation tolerance. Philosophical Transactions of the Royal Society of London. Series B, Biological Sciences. May 2001, 356 (1409): 617–23. PMC 1088449
 . PMID 11375065. doi:10.1098/rstb.2001.0845.
. PMID 11375065. doi:10.1098/rstb.2001.0845.
- ^ Halloran PF, Chang J, Famulski K, Hidalgo LG, Salazar ID, Merino Lopez M, Matas A, Picton M, de Freitas D, Bromberg J, Serón D, Sellarés J, Einecke G, Reeve J. Disappearance of T Cell-Mediated Rejection Despite Continued Antibody-Mediated Rejection in Late Kidney Transplant Recipients. Journal of the American Society of Nephrology. July 2015, 26 (7): 1711–20. PMC 4483591
 . PMID 25377077. doi:10.1681/ASN.2014060588.
. PMID 25377077. doi:10.1681/ASN.2014060588.
- ^ Steger U, Denecke C, Sawitzki B, Karim M, Jones ND, Wood KJ. Exhaustive differentiation of alloreactive CD8+ T cells: critical for determination of graft acceptance or rejection (PDF). Transplantation. May 2008, 85 (9): 1339–47 [2020-02-23]. PMID 18475193. doi:10.1097/TP.0b013e31816dd64a. (原始內容存檔 (PDF)於2020-02-23).
- ^ de Mare-Bredemeijer EL, Shi XL, Mancham S, van Gent R, van der Heide-Mulder M, de Boer R, Heemskerk MH, de Jonge J, van der Laan LJ, Metselaar HJ, Kwekkeboom J. Cytomegalovirus-Induced Expression of CD244 after Liver Transplantation Is Associated with CD8+ T Cell Hyporesponsiveness to Alloantigen. Journal of Immunology. August 2015, 195 (4): 1838–48. PMID 26170387. doi:10.4049/jimmunol.1500440.
- ^ Gassa A, Jian F, Kalkavan H, Duhan V, Honke N, Shaabani N, Friedrich SK, Dolff S, Wahlers T, Kribben A, Hardt C, Lang PA, Witzke O, Lang KS. IL-10 Induces T Cell Exhaustion During Transplantation of Virus Infected Hearts. Cellular Physiology and Biochemistry. 2016, 38 (3): 1171–81. PMID 26963287. doi:10.1159/000443067 (英語).
- ^ Shi XL, de Mare-Bredemeijer EL, Tapirdamaz Ö, Hansen BE, van Gent R, van Campenhout MJ, Mancham S, Litjens NH, Betjes MG, van der Eijk AA, Xia Q, van der Laan LJ, de Jonge J, Metselaar HJ, Kwekkeboom J. CMV Primary Infection Is Associated With Donor-Specific T Cell Hyporesponsiveness and Fewer Late Acute Rejections After Liver Transplantation. American Journal of Transplantation. September 2015, 15 (9): 2431–42. PMID 25943855. doi:10.1111/ajt.13288.
- ^ Woo SR, Turnis ME, Goldberg MV, Bankoti J, Selby M, Nirschl CJ, Bettini ML, Gravano DM, Vogel P, Liu CL, Tangsombatvisit S, Grosso JF, Netto G, Smeltzer MP, Chaux A, Utz PJ, Workman CJ, Pardoll DM, Korman AJ, Drake CG, Vignali DA. Immune inhibitory molecules LAG-3 and PD-1 synergistically regulate T-cell function to promote tumoral immune escape. Cancer Research. February 2012, 72 (4): 917–27. PMC 3288154
 . PMID 22186141. doi:10.1158/0008-5472.CAN-11-1620.
. PMID 22186141. doi:10.1158/0008-5472.CAN-11-1620.
- ^ Zelle-Rieser C, Thangavadivel S, Biedermann R, Brunner A, Stoitzner P, Willenbacher E, Greil R, Jöhrer K. T cells in multiple myeloma display features of exhaustion and senescence at the tumor site. Journal of Hematology & Oncology. November 2016, 9 (1): 116. PMC 5093947
 . PMID 27809856. doi:10.1186/s13045-016-0345-3 (英語).
. PMID 27809856. doi:10.1186/s13045-016-0345-3 (英語).
- ^ Lakins MA, Ghorani E, Munir H, Martins CP, Shields JD. + T Cells to protect tumour cells. Nature Communications. March 2018, 9 (1): 948. PMC 5838096
 . PMID 29507342. doi:10.1038/s41467-018-03347-0.
. PMID 29507342. doi:10.1038/s41467-018-03347-0.
- ^ Conforti, Laura. The ion channel network in T lymphocytes, a target for immunotherapy. Clinical Immunology. 2012-02-10, 142 (2): 105–106 [2020-02-23]. doi:10.1016/j.clim.2011.11.009. (原始內容存檔於2020-03-18) (英語).
- ^ Liu L, Chang YJ, Xu LP, Zhang XH, Wang Y, Liu KY, Huang XJ. T cell exhaustion characterized by compromised MHC class I and II restricted cytotoxic activity associates with acute B lymphoblastic leukemia relapse after allogeneic hematopoietic stem cell transplantation. Clinical Immunology. May 2018, 190: 32–40. PMID 29477343. doi:10.1016/j.clim.2018.02.009.
- ^ Kong Y, Zhang J, Claxton DF, Ehmann WC, Rybka WB, Zhu L, Zeng H, Schell TD, Zheng H. PD-1(hi)TIM-3(+) T cells associate with and predict leukemia relapse in AML patients post allogeneic stem cell transplantation. Blood Cancer Journal. July 2015, 5 (7): e330. PMC 4526784
 . PMID 26230954. doi:10.1038/bcj.2015.58 (英語).
. PMID 26230954. doi:10.1038/bcj.2015.58 (英語).
- ^ U.S. FDA Approved Immune-Checkpoint Inhibitors and Immunotherapies. Medical Writer Agency | 香港醫學作家 | MediPR | MediPaper Hong Kong. 2018-08-21 [2018-09-22]. (原始內容存檔於2018-09-04) (英國英語).
- ^ Bhadra R, Gigley JP, Weiss LM, Khan IA. Control of Toxoplasma reactivation by rescue of dysfunctional CD8+ T-cell response via PD-1-PDL-1 blockade. Proceedings of the National Academy of Sciences of the United States of America. May 2011, 108 (22): 9196–201. PMC 3107287
 . PMID 21576466. doi:10.1073/pnas.1015298108.
. PMID 21576466. doi:10.1073/pnas.1015298108.
外部連結[編輯]
- Immunobiology, 5th Edition(頁面存檔備份,存於網際網路檔案館)
- niaid.nih.gov – The Immune System
- T-cell Group – Cardiff University
- (Successful!) Treatment of Metastatic Melanoma with Autologous CD4+ T Cells against NY-ESO-1(頁面存檔備份,存於網際網路檔案館).
- The Center for Modeling Immunity to Enteric Pathogens (MIEP)(頁面存檔備份,存於網際網路檔案館)
- Anthony J. Davies. The tale of T cells. Immunology Today: 137–140. [2018-04-02]. doi:10.1016/0167-5699(93)90216-8. (原始內容存檔於2018-06-29).
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
