乳糜泻
此條目需要擴充。 (2016年12月22日) |
| 乳糜泻 Coeliac disease | |
|---|---|
| 又称 | 麦胶性肠病(gluten enteropathy)、非熱帶脂肪瀉(non-tropical steatorrhea) |
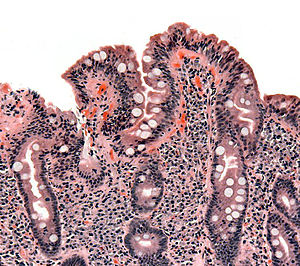 | |
| 乳糜泻患者的小肠絨毛切片,顯示小腸絨毛消失,陷窩肥厚且淋巴細胞浸潤。 | |
| 读音 | |
| 症状 | 无症状或非特异性症状、腹胀、腹泻、便秘、吸收不良、体重下降、疱疹性皮膚炎[1][2] |
| 併發症 | 缺铁性贫血、骨质疏松症、不孕、癌症、神经系统疾病、其他自體免疫性疾病[3][4][5][6][7] |
| 起病年龄 | 任何年齡[1][8] |
| 病程 | 終身[6] |
| 类型 | autoimmune disease of gastrointestinal tract[*]、麩質相關疾病[*]、疾病 |
| 病因 | 对麸质的反应[9] |
| 診斷方法 | 家族病史、血液抗体检测、肠道活检、基因检测、对移除麸质的反应[10][11] |
| 鑑別診斷 | 炎症性肠病、肠道寄生虫、腸激躁症、囊肿性纤维化[12] |
| 治療 | 无麸质饮食[13] |
| 患病率 | 约为1/135[14] |
| 分类和外部资源 | |
| 醫學專科 | 胃肠学、內科學 |
| ICD-11 | DA95 |
| ICD-9-CM | 579.0 |
| OMIM | 609754、612008、612005、612006、607202、611598、612007、612011、612009 |
| DiseasesDB | 2922 |
| MedlinePlus | 000233 |
| eMedicine | 932104、373864 |
| Orphanet | 555 |
乳糜瀉(英語:coeliac disease 或 celiac disease)又称麸质敏感性肠病(gluten-sensitive enteropathy)[15],是具有遗传倾向,对含麦胶食物不耐受而导致的肠源性吸收障碍的小肠自體免疫性疾病[10][16]。
典型的症狀包含胃腸道症狀,像是慢性腹瀉、腹脹、吸收不良、降低食慾,以及使孩童生長遲緩。這個病通常發生在六個月大到兩歲之間[1]。不典型的症狀比較常見,尤其是病人年紀大於兩歲時[8][17][18]。腸胃道症狀可能輕微或沒有表現,另外也可能會有許多症狀影響到身體任何部位,或甚至是沒有顯著症狀表現[1]。乳糜瀉剛開始是被發現於孩童身上[8][6],但其實任意年紀都可以發病[1][8]。此病常常和其他自體免疫性疾病共病,如1型糖尿病和甲狀腺炎等[6]。
机制
[编辑]乳糜瀉是因人體不適應麩質進而引發過敏反應的結果。麩質是小麥、大麥及裸麥等穀物含有的一組蛋白質[19][20][21]。一般而言,適量的食用燕麥若無與摻混其他含有麩質的穀物,通常不會對患者造成影響[20][22],而食用後會出現乳糜瀉問題的機率則可能與於燕麥的品種有關[20][23]。
此症是遺傳性疾病[24],患者攝取到麩質後,身體裡的異常免疫系統對此產生反應,並可能導致生成數種自身抗體而影響許多不同的器官[25][26]。在小腸中,自身抗體會引發炎症反應,並可能造成生長於小腸內壁的絨毛長度變短(絨毛萎縮)[24][27],而這會影響營養吸收,患者因而經常貧血[24][21]。
诊断
[编辑]确诊通常非常困难,大部分患者在正确诊断前都经歷较长的时间[28]。现在有多个检测手段可以使用。患者症状的严重程度可能决定了这些检测的顺序,但如果患者已经在进行无麸质饮食,则所有上述的检测都无效。小肠的损伤通常在饮食中去掉麸质后几周开始恢复,而抗体的水平也在数月后下降。对于那些正在进行无麸质饮食的人群,可能需要在其饮食中每日有一餐加入含麸质的食物,持续六周后再进行检测。[29]
診斷一般是透過血液抗體測試及腸道活體組織切片進行,會用特殊的基因檢測作為輔助[10]。不過診斷不容易直接進行[28],多半血液中的抗體檢驗是陰性的[30][31],而腸道健康的絨毛也只有少許變化[32]。病患在確診前多半已有嚴重症狀,且持續了幾年[33][34]。目前越來越多的確診案例來自於無症狀患者的篩檢結果[35]。不過目前尚無足夠實證佐證篩檢的效果[36]。此病症是由對於麩質蛋白質的永久不耐所造成[10],和更罕見的小麥過敏不同[37]。
治疗
[编辑]目前認為對此病唯一有效的治療方式是讓病患採取嚴格的終生無麩質飲食。這樣可讓大多數的患者腸粘膜復元、改善症狀並降低產生併發症的風險[13]。如果不加以治療,此病將可能導致如腸道淋巴瘤等癌症,並會略為增加早期死亡的風險[3]。世界各地罹患此病的人口比率各不相同,從1/300人至1/40不等,平均則是約在1/100至1/170間[14]。據估計,罹有此病者中有高達80%並未被確診出來,通常是因為他們的胃腸道症狀極為輕微或根本沒有出現症狀,以及因為大眾缺乏對於症狀和診斷標準的知識所致[5][33][38]。統計上,此病的女性患者較男性略多一些[39]。
参考文献
[编辑]- ^ 1.0 1.1 1.2 1.3 1.4 Fasano A. Clinical presentation of celiac disease in the pediatric population. Gastroenterology (Review). April 2005, 128 (4 Suppl 1): S68–73. PMID 15825129. doi:10.1053/j.gastro.2005.02.015.
- ^ Symptoms & Causes of Celiac Disease | NIDDK. National Institute of Diabetes and Digestive and Kidney Diseases. June 2016 [24 April 2017]. (原始内容存档于2017-04-24). (页面存档备份,存于互联网档案馆)
- ^ 3.0 3.1 Lebwohl B, Ludvigsson JF, Green PH. Celiac disease and non-celiac gluten sensitivity. BMJ (Review). October 2015, 351: h4347. PMC 4596973
 . PMID 26438584. doi:10.1136/bmj.h4347.
. PMID 26438584. doi:10.1136/bmj.h4347. Celiac disease occurs in about 1% of the population worldwide, although most people with the condition are undiagnosed. It can cause a wide variety of symptoms, both intestinal and extra-intestinal because it is a systemic autoimmune disease that is triggered by dietary gluten. Patients with coeliac disease are at increased risk of cancer, including a twofold to fourfold increased risk of non-Hodgkin’s lymphoma and a more than 30-fold increased risk of small intestinal adenocarcinoma, and they have a 1.4-fold increased risk of death.
- ^ 引用错误:没有为名为
Lund2015的参考文献提供内容 - ^ 5.0 5.1 Celiac disease. World Gastroenterology Organisation Global Guidelines. July 2016 [23 April 2017]. (原始内容存档于2017-03-17).
- ^ 6.0 6.1 6.2 6.3 Ciccocioppo R, Kruzliak P, Cangemi GC, Pohanka M, Betti E, Lauret E, Rodrigo L. The Spectrum of Differences between Childhood and Adulthood Celiac Disease. Nutrients (Review). 22 October 2015, 7 (10): 8733–51. PMC 4632446
 . PMID 26506381. doi:10.3390/nu7105426.
. PMID 26506381. doi:10.3390/nu7105426. Several additional studies in extensive series of coeliac patients have clearly shown that TG2A sensitivity varies depending on the severity of duodenal damage, and reaches almost 100% in the presence of complete villous atrophy (more common in children under three years), 70% for subtotal atrophy, and up to 30% when only an increase in IELs is present. (IELs: intraepithelial lymphocytes)
- ^ Lionetti E, Francavilla R, Pavone P, Pavone L, Francavilla T, Pulvirenti A, Giugno R, Ruggieri M. The neurology of coeliac disease in childhood: what is the evidence? A systematic review and meta-analysis. Developmental Medicine and Child Neurology. August 2010, 52 (8): 700–7. PMID 20345955. doi:10.1111/j.1469-8749.2010.03647.x
 .
.
- ^ 8.0 8.1 8.2 8.3 Husby S, Koletzko S, Korponay-Szabó IR, Mearin ML, Phillips A, Shamir R, Troncone R, Giersiepen K, Branski D, Catassi C, Lelgeman M, Mäki M, Ribes-Koninckx C, Ventura A, Zimmer KP, (( ESPGHAN Working Group on Coeliac Disease Diagnosis; ESPGHAN Gastroenterology Committee; European Society for Pediatric Gastroenterology, Hepatology, and Nutrition)). European Society for Pediatric Gastroenterology, Hepatology, and Nutrition guidelines for the diagnosis of coeliac disease (PDF). J Pediatr Gastroenterol Nutr (Practice Guideline). January 2012, 54 (1): 136–60 [2018-03-29]. PMID 22197856. doi:10.1097/MPG.0b013e31821a23d0. (原始内容 (PDF)存档于2016-04-03).
Since 1990, the understanding of the pathological processes of CD has increased enormously, leading to a change in the clinical paradigm of CD from a chronic, gluten-dependent enteropathy of childhood to a systemic disease with chronic immune features affecting different organ systems. (...) atypical symptoms may be considerably more common than classic symptoms
温哥华格式错误 (帮助) - ^ 引用错误:没有为名为
TovoliMasi2015的参考文献提供内容 - ^ 10.0 10.1 10.2 10.3 Celiac Disease. NIDDKD. June 2015 [17 March 2016]. (原始内容存档于2016-06-16).
- ^ 引用错误:没有为名为
VivasVaquero2015的参考文献提供内容 - ^ Ferri, Fred F. Ferri's differential diagnosis : a practical guide to the differential diagnosis of symptoms, signs, and clinical disorders 2nd. Philadelphia, PA: Elsevier/Mosby. 2010: Chapter C. ISBN 978-0323076999.
- ^ 13.0 13.1 See JA, Kaukinen K, Makharia GK, Gibson PR, Murray JA. Practical insights into gluten-free diets. Nat Rev Gastroenterol Hepatol (Review). 2015年10月, 12 (10): 580–91. PMID 26392070. doi:10.1038/nrgastro.2015.156.
A lack of symptoms and/or negative serological markers are not reliable indicators of mucosal response to the diet. Furthermore, up to 30% of patients continue to have gastrointestinal symptoms despite a strict GFD.122,124 If adherence is questioned, a structured interview by a qualified dietitian can help to identify both intentional and inadvertent sources of gluten.
- ^ 14.0 14.1 Fasano A, Catassi C. Clinical practice. Celiac disease. The New England Journal of Medicine (Review). December 2012, 367 (25): 2419–26. PMID 23252527. doi:10.1056/NEJMcp1113994.
- ^ Nelsen DA Jr. Gluten-sensitive enteropathy (celiac disease): more common than you think. Am Fam Physician. 2002;66(12):2259-2266.
- ^ Uche-Anya E, Lebwohl B. Celiac disease: clinical update. Curr Opin Gastroenterol. 2021;37(6):619-624. doi:10.1097/MOG.0000000000000785
- ^ Newnham, Evan D. Coeliac disease in the 21st century: Paradigm shifts in the modern age. Journal of Gastroenterology and Hepatology. 2017, 32: 82–85. PMID 28244672. doi:10.1111/jgh.13704.
Presentation of CD with malabsorptive symptoms or malnutrition is now the exception rather than the rule.

- ^ Tonutti E, Bizzaro N. Diagnosis and classification of celiac disease and gluten sensitivity. Autoimmun Rev. 2014, 13 (4–5): 472–6. PMID 24440147. doi:10.1016/j.autrev.2014.01.043.
- ^ Tovoli F, Masi C, Guidetti E, Negrini G, Paterini P, Bolondi L. Clinical and diagnostic aspects of gluten related disorders. World Journal of Clinical Cases (Review). March 2015, 3 (3): 275–84. PMC 4360499
 . PMID 25789300. doi:10.12998/wjcc.v3.i3.275.
. PMID 25789300. doi:10.12998/wjcc.v3.i3.275.
- ^ 20.0 20.1 20.2 Penagini F, Dilillo D, Meneghin F, Mameli C, Fabiano V, Zuccotti GV. Gluten-free diet in children: an approach to a nutritionally adequate and balanced diet. Nutrients (Review). November 2013, 5 (11): 4553–65. PMC 3847748
 . PMID 24253052. doi:10.3390/nu5114553.
. PMID 24253052. doi:10.3390/nu5114553.
- ^ 21.0 21.1 Di Sabatino A, Corazza GR. Coeliac disease. Lancet. April 2009, 373 (9673): 1480–93. PMID 19394538. doi:10.1016/S0140-6736(09)60254-3.
- ^ Pinto-Sánchez MI, Causada-Calo N, Bercik P, Ford AC, Murray JA, Armstrong D, Semrad C, Kupfer SS, Alaedini A, Moayyedi P, Leffler DA, Verdú EF, Green P. Safety of Adding Oats to a Gluten-Free Diet for Patients With Celiac Disease: Systematic Review and Meta-analysis of Clinical and Observational Studies (PDF). Gastroenterology. August 2017, 153 (2): 395–409.e3 [2020-02-20]. PMID 28431885. doi:10.1053/j.gastro.2017.04.009. (原始内容存档 (PDF)于2021-01-29).
- ^ Comino I, Moreno M, Sousa C. Role of oats in celiac disease. World Journal of Gastroenterology. November 2015, 21 (41): 11825–31. PMC 4631980
 . PMID 26557006. doi:10.3748/wjg.v21.i41.11825.
. PMID 26557006. doi:10.3748/wjg.v21.i41.11825. It is necessary to consider that oats include many varieties, containing various amino acid sequences and showing different immunoreactivities associated with toxic prolamins. As a result, several studies have shown that the immunogenicity of oats varies depending on the cultivar consumed. Thus, it is essential to thoroughly study the variety of oats used in a food ingredient before including it in a gluten-free diet.
- ^ 24.0 24.1 24.2 Celiac Disease. NIDDKD. June 2015 [17 March 2016]. (原始内容存档于2016-06-16).
- ^ Lundin KE, Wijmenga C. Coeliac disease and autoimmune disease-genetic overlap and screening. Nature Reviews. Gastroenterology & Hepatology (Review). September 2015, 12 (9): 507–15. PMID 26303674. doi:10.1038/nrgastro.2015.136.
The abnormal immunological response elicited by gluten-derived proteins can lead to the production of several different autoantibodies, which affect different systems.
- ^ National Institute for Health and Clinical Excellence. Clinical guideline 86: Recognition and assessment of coeliac disease. London, 2015.
- ^ Vivas S, Vaquero L, Rodríguez-Martín L, Caminero A. Age-related differences in celiac disease: Specific characteristics of adult presentation. World Journal of Gastrointestinal Pharmacology and Therapeutics (Review). November 2015, 6 (4): 207–12. PMC 4635160
 . PMID 26558154. doi:10.4292/wjgpt.v6.i4.207.
. PMID 26558154. doi:10.4292/wjgpt.v6.i4.207. In addition, the presence of intraepithelial lymphocytosis and/or villous atrophy and crypt hyperplasia of small-bowel mucosa, and clinical remission after withdrawal of gluten from the diet, are also used for diagnosis antitransglutaminase antibody (tTGA) titers and the degree of histological lesions inversely correlate with age. Thus, as the age of diagnosis increases antibody titers decrease and histological damage is less marked. It is common to find adults without villous atrophy showing only an inflammatory pattern in duodenal mucosa biopsies: Lymphocytic enteritis (Marsh I) or added crypt hyperplasia (Marsh II)
- ^ 28.0 28.1 Matthias T, Pfeiffer S, Selmi C, Eric Gershwin M. Diagnostic challenges in celiac disease and the role of the tissue transglutaminase-neo-epitope. Clin Rev Allergy Immunol (Review). 2010年4月, 38 (2–3): 298–301. PMID 19629760. doi:10.1007/s12016-009-8160-z.
- ^ National Institute for Health and Clinical Excellence. Clinical guideline 86: Recognition and assessment of coeliac disease. London, 2015.
- ^ Lewis NR, Scott BB. Systematic review: the use of serology to exclude or diagnose coeliac disease (a comparison of the endomysial and tissue transglutaminase antibody tests). Alimentary Pharmacology & Therapeutics. July 2006, 24 (1): 47–54. PMID 16803602. doi:10.1111/j.1365-2036.2006.02967.x.
- ^ Rostom A, Murray JA, Kagnoff MF. American Gastroenterological Association (AGA) Institute technical review on the diagnosis and management of celiac disease. Gastroenterology (Review). December 2006, 131 (6): 1981–2002 [2022-07-20]. PMID 17087937. doi:10.1053/j.gastro.2006.10.004. (原始内容存档于2014-03-18).
- ^ Molina-Infante J, Santolaria S, Sanders DS, Fernández-Bañares F. Systematic review: noncoeliac gluten sensitivity. Alimentary Pharmacology & Therapeutics (Review). May 2015, 41 (9): 807–20. PMID 25753138. doi:10.1111/apt.13155.
Furthermore, seronegativity is more common in coeliac disease patients without villous atrophy (Marsh 1-2 lesions), but these ‘minor’ forms of coeliac disease may have similar clinical manifestations to those with villous atrophy and may show similar clinical–histological remission with reversal of haematological or biochemical disturbances on a gluten-free diet (GFD).
- ^ 33.0 33.1 Cichewicz AB, Mearns ES, Taylor A, Boulanger T, Gerber M, Leffler DA, et al. Diagnosis and Treatment Patterns in Celiac Disease. Dig Dis Sci (Review). 1 March 2019, 64 (8): 2095–2106. PMID 30820708. doi:10.1007/s10620-019-05528-3.
- ^ Ludvigsson JF, Card T, Ciclitira PJ, Swift GL, Nasr I, Sanders DS, Ciacci C. Support for patients with celiac disease: A literature review. United European Gastroenterology Journal (Review). April 2015, 3 (2): 146–59. PMC 4406900
 . PMID 25922674. doi:10.1177/2050640614562599.
. PMID 25922674. doi:10.1177/2050640614562599.
- ^ van Heel DA, West J. Recent advances in coeliac disease. Gut (Review). July 2006, 55 (7): 1037–46. PMC 1856316
 . PMID 16766754. doi:10.1136/gut.2005.075119.
. PMID 16766754. doi:10.1136/gut.2005.075119.
- ^ Bibbins-Domingo K, Grossman DC, Curry SJ, Barry MJ, Davidson KW, Doubeni CA, Ebell M, Epling JW, Herzstein J, Kemper AR, Krist AH, Kurth AE, Landefeld CS, Mangione CM, Phipps MG, Silverstein M, Simon MA, Tseng CW. Screening for Celiac Disease: US Preventive Services Task Force Recommendation Statement. JAMA. March 2017, 317 (12): 1252–1257. PMID 28350936. doi:10.1001/jama.2017.1462.
- ^ Burkhardt, J. G.; Chapa-Rodriguez, A.; Bahna, S. L. Gluten sensitivities and the allergist: Threshing the grain from the husks. Allergy. July 2018, 73 (7): 1359–1368. PMID 29131356. doi:10.1111/all.13354.
- ^ Lionetti E, Gatti S, Pulvirenti A, Catassi C. Celiac disease from a global perspective. Best Practice & Research. Clinical Gastroenterology (Review). June 2015, 29 (3): 365–79. PMID 26060103. doi:10.1016/j.bpg.2015.05.004.
- ^ Hischenhuber C, Crevel R, Jarry B, Mäki M, Moneret-Vautrin DA, Romano A, Troncone R, Ward R. Review article: safe amounts of gluten for patients with wheat allergy or coeliac disease. Alimentary Pharmacology & Therapeutics. March 2006, 23 (5): 559–75. PMID 16480395. doi:10.1111/j.1365-2036.2006.02768.x.
